
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 26E
Consider the following bases: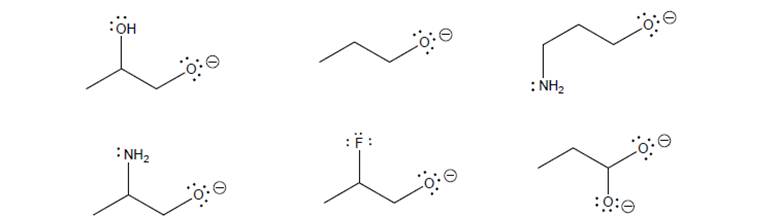
a. For each base above, circle the atom/atoms with the highest PE (will release the most P.E.when a lone pair on this atom combines with an
b. Rank the bases 1 (highest P.E./strongest base) to 7 (lowest PE/weakest base), and explainyour reasoning.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Label all of the acidic hydrogen. (Do not count any of the ones that are not shown)
bns 2001320
Circle the one that is the most acidic
kne noltamoino siano ws
Explain, using CARDIO, why it is the most acidic.
H
H
H
H
N.
H.
H
H
stasib was baie vincul
CI
6057 enim09
ogmo) ons nod:6) istim relev
H
S
WH bus il basi
WH Uns 2506 A to
Rank the acids in the table below from strongest (1) to weakest (4). The most acidic H atom in each acd has been highlighted.
H.
H.
H
(Choose one)
(Choose one)
(Choose one)
(Choose one)
The strength of Lewis bases follows the order of...
A. NH3 > Me2NH > Me3N
B. Me3N > Me2NH > MeNH2
C. NH3 > MeNH2 > EtNH2
D. Me2NH > MeNH2 > NH3
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 4 - Prob. 1CTQCh. 4 - Figure 4.1 is a cartoon depiction of liquid water...Ch. 4 - Prob. 3CTQCh. 4 - Prob. 4CTQCh. 4 - In HF , neither H nor F holds a full formal charge...Ch. 4 - Prob. 6CTQCh. 4 - Prob. 7CTQCh. 4 - Prob. 8CTQCh. 4 - Within any one section of Table 4.2, boiling...Ch. 4 - Prob. 10CTQ
Ch. 4 - Prob. 11CTQCh. 4 - Prob. 12CTQCh. 4 - Prob. 13CTQCh. 4 - Prob. 14CTQCh. 4 - Prob. 15CTQCh. 4 - Prob. 16CTQCh. 4 - Prob. 17CTQCh. 4 - Prob. 18CTQCh. 4 - Prob. 19CTQCh. 4 - Prob. 20CTQCh. 4 - Prob. 21CTQCh. 4 - Prob. 22CTQCh. 4 - (E) Label each of the following as strong acid,...Ch. 4 - Prob. 24CTQCh. 4 - Draw the structure of the conjugate base of water....Ch. 4 - Does Cl have a conjugate acid? If so, what is it?...Ch. 4 - Draw the conjugate base of CH4 (methane).Ch. 4 - For the previous four questions, label each...Ch. 4 - Prob. 29CTQCh. 4 - According to the conventions above, what is the...Ch. 4 - Draw an arrow on Figure 4.13 representing Hrxn4 ....Ch. 4 - Prob. 32CTQCh. 4 - Add a + or above each curved arrow in Figure 4.11...Ch. 4 - Prob. 34CTQCh. 4 - Prob. 35CTQCh. 4 - Prob. 36CTQCh. 4 - Prob. 37CTQCh. 4 - Prob. 38CTQCh. 4 - Prob. 39CTQCh. 4 - Prob. 40CTQCh. 4 - Prob. 41CTQCh. 4 - Prob. 42CTQCh. 4 - Prob. 43CTQCh. 4 - Prob. 44CTQCh. 4 - Prob. 45CTQCh. 4 - Prob. 46CTQCh. 4 - For NH3 (ammonia) and H2O (water)... a. Use curved...Ch. 4 - Prob. 48CTQCh. 4 - Prob. 49CTQCh. 4 - Prob. 50CTQCh. 4 - Prob. 51CTQCh. 4 - Prob. 52CTQCh. 4 - Prob. 53CTQCh. 4 - Prob. 1ECh. 4 - Prob. 2ECh. 4 - Prob. 3ECh. 4 - Prob. 4ECh. 4 - Prob. 5ECh. 4 - Prob. 6ECh. 4 - Prob. 7ECh. 4 - Prob. 8ECh. 4 - Propanal (bp 48°C) and propanol (bp 97°C), both...Ch. 4 - Rank the following molecules from lowest to...Ch. 4 - Prob. 12ECh. 4 - For each molecule below, draw the conjugate acid...Ch. 4 - For each structure you drew in the answer to the...Ch. 4 - Mark each of the following statements True or...Ch. 4 - Organic chemistry is a bit like cooking. Later in...Ch. 4 - Prob. 17ECh. 4 - Prob. 18ECh. 4 - Are endothermic reactions favorable or...Ch. 4 - Prob. 20ECh. 4 - Is bond formation endothermic or exothermic? Write...Ch. 4 - Summarize the relationship between pKa and acid...Ch. 4 - Summarize the relationship between pKa and base...Ch. 4 - Prob. 25ECh. 4 - Consider the following bases: a. For each base...Ch. 4 - Prob. 27ECh. 4 - The following are equivalent ways of asking about...Ch. 4 - Prob. 29E
Additional Science Textbook Solutions
Find more solutions based on key concepts
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
covered a synthesis of alkynes by a double dehydrohalogenation of dihalides. A student tried to convert trans-2...
Organic Chemistry (9th Edition)
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (5th Edition) (Standalone Book)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The following are equivalent ways of asking about the acidity of an H atom: • What is the most acidic H on the molecule? • Which H is associated with the published pKa value? • Which H on the molecule is easiest to remove? • Which H on the molecule takes the least energy to remove? • Which bond to an H is most polarized? • For which H atom is removal least uphill in energy? • Which bond to an H atom, when broken, results in the lowest PE conjugate base? We will often find the last of these questions is easiest to answer. To do this, find all the different Hatoms on the molecule, and draw all possible conjugate bases.Only the lowest-energy one is the “real” conjugate base. Identify this structure, and you have found the most acidic H. Use this strategy to find the most acidic H on each of the following molecules. Note: Each structure hasat least three different kinds of H’s, so draw at least three unique conjugate bases for each.arrow_forwardPhenol (shown below) has a pKa10 . a. Based on pKa data, is phenol a stronger or weaker acid than an ordinaryalcohol (e.g.. CH3OH ) or water? b. Draw the conjugate base of phenol (called phenoxide) including allimportant resonance structures. c. Construct an explanation for why phenol is a stronger acid than anordinary alcohol. (You may want to consider first why phenoxide is lowerin PE than methoxide (RO) or hydroxide (HO) .arrow_forwardFor each molecule below, draw the conjugate acid or conjugate base or both if the molecule hasboth a conjugate acid and a conjugate base (e.g., water).arrow_forward
- Complete the equation for the reaction between each Lewis acid-base pair. In each equation, label which starting material is the Lewis acid and which is the Lewis base; use curved arrows to show the flow of electrons in each reaction. In doing this problem, it is essential that you show valence electrons for all atoms participating in each reaction. (a) (b) (c) (d)arrow_forwardWhich of the ff. acids is weaker: HBrO4 or HIO4? Type your answer. (Note: Case, Space, & Format sensitive)arrow_forward5. In each pair, which species is a stronger base? Justify your answer. Farrow_forward
- 2. Explain the following trend (Note: "smaller pka-more acidic" is not an explanation, its a definition. Explanations require "why.") H pKa H O 9.95 H PIILO 41 H H CH H 50arrow_forwardAmong three bases, X, Y¯, and Z¯, the strongest one is Y¯, and the weakest one is Z-. Rank their conjugate acids, HX, HY, and HZ, in order of decreasing strength. Rank the acids from strongest to weakest. To rank items as equivalent, overlap them. • View Available Hint(s) Reset Help HZ HY HX Strongest acid Weakest acidarrow_forwardAmong three bases, X, Y¯, and Z¯, the strongest one is Y , and the weakest one is Z. Rank their conjugate acids, H HY, and HZ, in order of decreasing strength. Rank the acids from strongest to weakest. To rank items as equivalent, overlap them. • View Available Hint(s) Reset Help HZ HY HX Strongest acid Weakest acidarrow_forward
- 1. HO. HN a. polar/non-polar; b. Neutral/acidic/basic c. Also write whether the side chain is Hydrophobic/Hydrophilic 2. H2N CH- он CH2 a. polar/non-polar; b. Neutral/acidic/basic c. Also write whether the side chain is Hydrophobic/Hydrophilicarrow_forward1.3. Using concepts learned from acid-base theory in class, choose species from the list below that would form strongest interaction with Na F- H- CN- 2 points 1.4. Using concepts learned from acid-base theory in class, choose species from the list below that would form strongest interaction with Au OH- NH3 NCS- O RSHarrow_forwardWhich of the following compounds is the stronger base? Explain. or NH3 is a v base than NCI3. This is due to the difference in the electron withdrawing strength of Cl compared with H. v is more electronegative than and will v more electron density v the nitrogen than Thus, the nitrogen atom in NCI3 has a v positive charge than it does in NH3. The v positive charge means that is v difficult to add H* to NCI3 than it is to add H* to NH3. Is the compound Sb(OH)5 likely to be acidic, basic, or neutral in water? Justify your answer. Phosphorus and arsenic form acids of the form X(OH)n, so one would predict that antimony would be similar as it is in the same family. Based in the number of OH groups present, it should be However, antimony hydroxides are not stable and form oxides which are known to be amphoteric. Thus, Sb(OH)6 is in fact more likely to bearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY