
Concept explainers
Interpretation: The weakest acid and weakest base in below table should be determined.
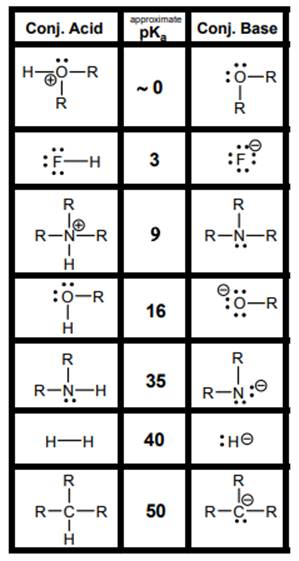
Concept introduction: According to Bronsted-Lowry concept, substance that donates proton is termed as acid while that accepts or gains protons is called base. Species formed after loss of protons from acids are known as their respective conjugate bases whereas conjugate acid is produced by addition of protons to base. Strength of conjugate acids and conjugate bases are inversely related to strengths of their respective bases and acids.
According to Lewis concept, substance that donates electron pair is termed as base while that accepts or gains electron pair is called acid. For example,
According to Arrhenius concept, substances that donate hydrogen ions in solutions are known as acids while bases are substances that release hydroxide ions in solutions.
Trending nowThis is a popular solution!

Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
- For NH3 (ammonia) and H2O (water)... a. Use curved arrows to show the most likely acid-base reaction, and draw the resultingproducts. (Hint: First decide which is the stronger acid, and which is the stronger base.) b. Mark each curved arrow with a positive (bond-breaking) or negative (bond-forming) numberindicating the energy change associated with that arrow (in pKa units). c. Calculate Hrxn and write this number above a set of reaction arrows that indicate whichdirection is downhill/favorable (in the example, the reaction is downhill to the right). d. Sketch an energy diagram for the reaction. e. Is your energy diagram consistent with the fact that, in this case, the most likely acid-basereaction is endothermic?arrow_forwardWater is considered the solvent of life. Water's polarity makes it unique, and this polarity is responsible for the life giving properties of water. In this short essay question, answer only one of the following questions, A OR B. In your first sentence, tell me if your answer is for A or B. NOTE that saying "water is polar, so it is cohesive", or "water is polar, so it moderates temperature", or "water is polar, so is more dense as liquid", with a description of how that life giving property is beneficial to life, does NOT answer this question! You must explain, in your OWN words, HOW the polarity of the water is mechanistically responsible for the life giving property you choose. Question B: Explain in detail how the polar nature of water allows fish to survive and swim around in the watery depths of Lake Baikal, Russia, all winter long in spite of the average air temperatures of minus 25 degrees Celsius in this frozen, snow covered region. What life giving property of water is…arrow_forwardWhich do you expect to be the stronger base: HCN or HNC? Explain. Hint: Draw out the complete Lewis structure for each molecule.arrow_forward
- 1. Determine how many types of nonequivalent protons are present in the following molecule. Assign letters for each kind of proton. а. b. N' d.arrow_forwardFor conjugate acid-base pair, identify the first species as an acid or a base and the second species as its conjugate base or conjugate acid. In addition, draw Lewis structures for each species, showing all valence electrons and any formal charges. Q) CH3NH2, CH3NH3+arrow_forwardWrite the net ionic chemical equilibrium that gets established formic acid, is dissolved in water. Use full lewis structures and show all lone electron pairs for reactants and products. Above each reaction arrow, write whether the reaction represents a Ka or Kb reaction.arrow_forward
- 2. Give the shape and the corresponding bond angle for a molecule of formula QX, where Q is an element from Group 4 of the periodic table. 3. State the shape of the PH, molecule and explain using electron-pair repulsion theory how this shape arises.arrow_forwardAssign which true or false statement: 1) Atoms with the largest electronegativity differences formed covalent bonds. 2) The trend that atoms like to have eight electrons in their valence shell called hexa rule 3) Passivation is a non-protective coating for metals. 4) Molten salts or solutions of salts conduct electricity 5) The oxides and hydroxides of gallium exhibit both acidic and basic behaviorsarrow_forward( do 2 with explanation)arrow_forward
- Draw a Lewis structure for the conjugate acid. Write in any non-zero formal charges and make sure all atoms in your structure fully satisfy the octet rule. molecule H :: H-C-Br: conjugate acid of the molecule Click and drag to start drawing a structure. 10 Xarrow_forwardProblem 4.18 Part A Arrange the elements commonly bonded to carbon in organic compounds, H, N, O, P, and S, in order of decreasing electronegativity. Rank the elements from strongest to weakest. To rank items as equivalent, overlap them. Most electronegative I OP Q Search Reset Help Least electronegative Pearson 4 Copyright © 2023 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Policy | Permissions | Caarrow_forwardCalculate Kb for each of the following ions: CN-, F-, CH3COO-, HCO3arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
