
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 4, Problem 29CTQ
Interpretation Introduction
Interpretation: Whether below reactions are endothermic or exothermic should be determined.
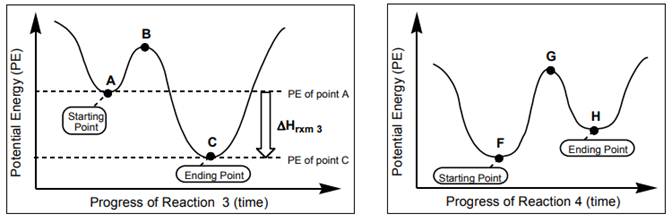
Concept introduction:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Question 5 of 8
O Macmillan Learning
>
Calculate AH° for the reaction using the given bond dissociation energies.
CH₂(g) + 2O₂(g) → CO₂(g) + 2 H₂O(g)
Bond AH° (kJ/mol)
O-O
H-O
C-H
C=O
O=O
C-O
ΔΗ° =
142
459
411
799
498
358
kJ/mol
This reaction is
exothermic.
endothermic.
Jorge is trying to classify the reaction represented by
the graph below and calculate the change in energy, AH
for it.
250
200-
..
PE
150
(KJ)
....
100
...
50
Reaction pathway
He claims that the AH will be -100kJ and the reaction is
exothermic. Is his claim correct? If not, find the errors
and fix them to give the correct answer. Make sure to
show ALL work and give explanations.
40 kJ
Reactants
Na
Activation Energy
CI
30 kJ
Products
Na
CI
H
10 kJ
Reaction Progress
Na
H CI
Potential Energy [kJ]
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 4 - Prob. 1CTQCh. 4 - Figure 4.1 is a cartoon depiction of liquid water...Ch. 4 - Prob. 3CTQCh. 4 - Prob. 4CTQCh. 4 - In HF , neither H nor F holds a full formal charge...Ch. 4 - Prob. 6CTQCh. 4 - Prob. 7CTQCh. 4 - Prob. 8CTQCh. 4 - Within any one section of Table 4.2, boiling...Ch. 4 - Prob. 10CTQ
Ch. 4 - Prob. 11CTQCh. 4 - Prob. 12CTQCh. 4 - Prob. 13CTQCh. 4 - Prob. 14CTQCh. 4 - Prob. 15CTQCh. 4 - Prob. 16CTQCh. 4 - Prob. 17CTQCh. 4 - Prob. 18CTQCh. 4 - Prob. 19CTQCh. 4 - Prob. 20CTQCh. 4 - Prob. 21CTQCh. 4 - Prob. 22CTQCh. 4 - (E) Label each of the following as strong acid,...Ch. 4 - Prob. 24CTQCh. 4 - Draw the structure of the conjugate base of water....Ch. 4 - Does Cl have a conjugate acid? If so, what is it?...Ch. 4 - Draw the conjugate base of CH4 (methane).Ch. 4 - For the previous four questions, label each...Ch. 4 - Prob. 29CTQCh. 4 - According to the conventions above, what is the...Ch. 4 - Draw an arrow on Figure 4.13 representing Hrxn4 ....Ch. 4 - Prob. 32CTQCh. 4 - Add a + or above each curved arrow in Figure 4.11...Ch. 4 - Prob. 34CTQCh. 4 - Prob. 35CTQCh. 4 - Prob. 36CTQCh. 4 - Prob. 37CTQCh. 4 - Prob. 38CTQCh. 4 - Prob. 39CTQCh. 4 - Prob. 40CTQCh. 4 - Prob. 41CTQCh. 4 - Prob. 42CTQCh. 4 - Prob. 43CTQCh. 4 - Prob. 44CTQCh. 4 - Prob. 45CTQCh. 4 - Prob. 46CTQCh. 4 - For NH3 (ammonia) and H2O (water)... a. Use curved...Ch. 4 - Prob. 48CTQCh. 4 - Prob. 49CTQCh. 4 - Prob. 50CTQCh. 4 - Prob. 51CTQCh. 4 - Prob. 52CTQCh. 4 - Prob. 53CTQCh. 4 - Prob. 1ECh. 4 - Prob. 2ECh. 4 - Prob. 3ECh. 4 - Prob. 4ECh. 4 - Prob. 5ECh. 4 - Prob. 6ECh. 4 - Prob. 7ECh. 4 - Prob. 8ECh. 4 - Propanal (bp 48°C) and propanol (bp 97°C), both...Ch. 4 - Rank the following molecules from lowest to...Ch. 4 - Prob. 12ECh. 4 - For each molecule below, draw the conjugate acid...Ch. 4 - For each structure you drew in the answer to the...Ch. 4 - Mark each of the following statements True or...Ch. 4 - Organic chemistry is a bit like cooking. Later in...Ch. 4 - Prob. 17ECh. 4 - Prob. 18ECh. 4 - Are endothermic reactions favorable or...Ch. 4 - Prob. 20ECh. 4 - Is bond formation endothermic or exothermic? Write...Ch. 4 - Summarize the relationship between pKa and acid...Ch. 4 - Summarize the relationship between pKa and base...Ch. 4 - Prob. 25ECh. 4 - Consider the following bases: a. For each base...Ch. 4 - Prob. 27ECh. 4 - The following are equivalent ways of asking about...Ch. 4 - Prob. 29E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The H0 and S0 for the reaction N2O4 N2 + 2O2?arrow_forwardD. H20 (s) –> H20 (I) AHo = 6.01kJ/mol| 1. This reaction depicts the 2. Is this reaction endothermic or exothermic? 3. Is this reaction reversible or irreversible? (process) of (product).arrow_forward(c) The energy of the reactants is shown on the following energy diagram. On the right side of the energy diagram, draw a horizontal line segment to indicate the energy of the products. Draw a vertical double-headed arrow (1) on the graph that corresponds to the value of AH for the reaction. CH4 + F2 Reaction Progress Energyarrow_forward
- Would the amount of heat absorbed by the dissolution in Example 5.6 appear greater, lesser, or remain the same if the experimenter used a calorimeter that was a poorer insulator than a coffee cup calorimeter? Explain your answer.arrow_forward7-16 A quart of milk quickly spoils if left at room temperature but keeps for several days in a refrigerator. Explain.arrow_forwardThe reaction 4 PH3 + 2 H2 + 9 O2 <=> 2 P2O5 + 8 H2O + 25 kJ is endothermic/exothermic (circle one) gives off heat/absorbs heat (circle one)arrow_forward
- 1. Fill in the boxes below with the correct reagent, reactant or product (5 (а) OH (b) 1. (CH),СHMgBr 2. H,0 ÓH (c) 1. L¡AIH, 2. НО (d) CH;OH H,SO4 (e) 1. 2 (CH3),CHMgBr, 2. H,0 ÓH (f) 1. LIAIH, 2. H,0 HO,arrow_forwardThis reaction is exothermic. Select one: True Falsearrow_forwardenergy (J) A B Iarrow_forward
- of CH,COOH Heat CO,Et ( 12 NaOE! CO,Et 2 Br но 1.Br PBr OH, OEt 1. NaOEt 2. NaOEt LDA CN CH,CH,a CH NaOH C, excessarrow_forwardWhich of the following reactions will release the most energy? O CH41) + O2(1) CO(g) + 2H20(g) O CHA(1) + O2() CO2() + 2H20(1) O CHA(g) + O2lg) CO2(g) + 2H2O(g) O CHA(g) + O2(g) CO2() + 2H2O(1)arrow_forwardCan you please help with reaction 2 and 3 under calculations?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax