
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 4, Problem 51CTQ
Interpretation Introduction
Interpretation: Reason for no dipole moment arrows for
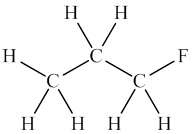
Concept introduction: Dipole moment is measure of bond polarity in molecule. It is defined as product of charge and distance of separation between opposite charges. Since dipole moment is result of charge separation, electronegativity difference plays significant role to govern dipole moment in molecules. Dipole moment exists for molecules that have significant amount of electronegativity difference between bonded atoms.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
please help - for the last drawing.. (second arrow down) - after this.. please help this answer
Could we cut just one bond in the "starting" molecule shown in the drawing area below to create this "target" molecule?
If so, highlight the bond to be cut. If not, check the box under the drawing area that says Not possible.
Note: it's OK if cutting the bond creates more than one molecule, as long as one of them is the target molecule.
Not possible.
The target molecule.
H
H
Note for advanced students: what we mean by "cutting" the bond here is breaking the bond and attaching H atoms to each dangling end, like this:
++*++
H
C-H H-ő-H
H
X
-Ö-H
S
The instructions are to draw the resonance hybrid structure of the molecule on the left. My answer is circled , which is wrong. Can someone explain why?
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 4 - Prob. 1CTQCh. 4 - Figure 4.1 is a cartoon depiction of liquid water...Ch. 4 - Prob. 3CTQCh. 4 - Prob. 4CTQCh. 4 - In HF , neither H nor F holds a full formal charge...Ch. 4 - Prob. 6CTQCh. 4 - Prob. 7CTQCh. 4 - Prob. 8CTQCh. 4 - Within any one section of Table 4.2, boiling...Ch. 4 - Prob. 10CTQ
Ch. 4 - Prob. 11CTQCh. 4 - Prob. 12CTQCh. 4 - Prob. 13CTQCh. 4 - Prob. 14CTQCh. 4 - Prob. 15CTQCh. 4 - Prob. 16CTQCh. 4 - Prob. 17CTQCh. 4 - Prob. 18CTQCh. 4 - Prob. 19CTQCh. 4 - Prob. 20CTQCh. 4 - Prob. 21CTQCh. 4 - Prob. 22CTQCh. 4 - (E) Label each of the following as strong acid,...Ch. 4 - Prob. 24CTQCh. 4 - Draw the structure of the conjugate base of water....Ch. 4 - Does Cl have a conjugate acid? If so, what is it?...Ch. 4 - Draw the conjugate base of CH4 (methane).Ch. 4 - For the previous four questions, label each...Ch. 4 - Prob. 29CTQCh. 4 - According to the conventions above, what is the...Ch. 4 - Draw an arrow on Figure 4.13 representing Hrxn4 ....Ch. 4 - Prob. 32CTQCh. 4 - Add a + or above each curved arrow in Figure 4.11...Ch. 4 - Prob. 34CTQCh. 4 - Prob. 35CTQCh. 4 - Prob. 36CTQCh. 4 - Prob. 37CTQCh. 4 - Prob. 38CTQCh. 4 - Prob. 39CTQCh. 4 - Prob. 40CTQCh. 4 - Prob. 41CTQCh. 4 - Prob. 42CTQCh. 4 - Prob. 43CTQCh. 4 - Prob. 44CTQCh. 4 - Prob. 45CTQCh. 4 - Prob. 46CTQCh. 4 - For NH3 (ammonia) and H2O (water)... a. Use curved...Ch. 4 - Prob. 48CTQCh. 4 - Prob. 49CTQCh. 4 - Prob. 50CTQCh. 4 - Prob. 51CTQCh. 4 - Prob. 52CTQCh. 4 - Prob. 53CTQCh. 4 - Prob. 1ECh. 4 - Prob. 2ECh. 4 - Prob. 3ECh. 4 - Prob. 4ECh. 4 - Prob. 5ECh. 4 - Prob. 6ECh. 4 - Prob. 7ECh. 4 - Prob. 8ECh. 4 - Propanal (bp 48°C) and propanol (bp 97°C), both...Ch. 4 - Rank the following molecules from lowest to...Ch. 4 - Prob. 12ECh. 4 - For each molecule below, draw the conjugate acid...Ch. 4 - For each structure you drew in the answer to the...Ch. 4 - Mark each of the following statements True or...Ch. 4 - Organic chemistry is a bit like cooking. Later in...Ch. 4 - Prob. 17ECh. 4 - Prob. 18ECh. 4 - Are endothermic reactions favorable or...Ch. 4 - Prob. 20ECh. 4 - Is bond formation endothermic or exothermic? Write...Ch. 4 - Summarize the relationship between pKa and acid...Ch. 4 - Summarize the relationship between pKa and base...Ch. 4 - Prob. 25ECh. 4 - Consider the following bases: a. For each base...Ch. 4 - Prob. 27ECh. 4 - The following are equivalent ways of asking about...Ch. 4 - Prob. 29E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Read This! The attractive and repulsive forces in an atom are rather complex. An electron is attracted to the protons in the nucleus, but it is also repelled by the other electrons in the atom. It is important to note however that the attractive force of the nucleus is NOT divided up among the electrons in the atom. Each electron gets approximately the full attractive force of the nucleus (minus the repulsive effects of other electrons). Compare the diagram below to set D in Model 3. Notice the similarity in attractive force. 0.10 nm 0.10 nm approx. 4.60 x 10-8 (on each electron) Model 4 – Period 3 Elements Aluminum Chlorine Sodium What does it MEAN? What do you WONDER? What do you SEE?arrow_forwardI'm very confused on this. please help thank youarrow_forwardConsider this Lewis structure in the curved arrows joke. Draw the resident structure obtained by moving electrons as indicated by the curved arrows.arrow_forward
- Q.e 3 please answer the question and make sure it is correct, thank you.arrow_forwardHaving toruble understand this one, wanted to see if i had it correct.arrow_forwardA. For the molecule circled, draw the potential energy using this diagram. B. Draw the other three Newman projections based on the drawing at the bottom leftarrow_forward
- The next part of the problem is to draw the resonance hybrid. (Exclude formal charges) I’m not sure where to put the dotted resonance lines of the hybrid structure.arrow_forwardx-xo B Draw molecule A. On that drawing include the lone pairs and the curved arrows that would produce resonance structure B.arrow_forwardDraw an exact 3-D representation of the molecule from the model. please answer all question step by step. Answer follow image:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning