
Concept explainers
Interpretation: Among below molecules, molecules that are capable of hydrogen bonding to them should be determined.
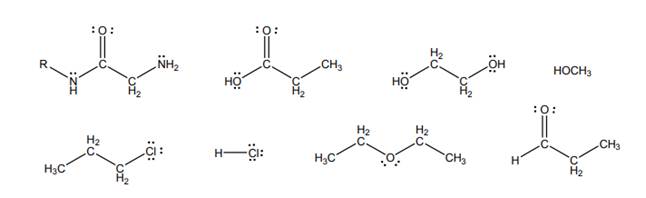
Concept introduction: Intermolecular forces are types of forces that are responsible to hold atoms together in molecule. There are various types of intermolecular forces as follows:
1. Hydrogen bonding:
As is evident from its name, these types of forces exist if hydrogen is present. Such forces are present when hydrogen atom bonds with highly electronegative elements like
2. Ion-dipole forces:
This force exists between ion and molecules with dipole moment in them. Attraction exists between ion and oppositely charged end of dipole.
3. Ion-induced dipole forces:
When an ion approaches non-polar molecule, it induces temporary dipole in it. Ion gets attracted towards oppositely charged part of dipole induced in molecule.
4. Dispersion forces:
These forces are also known as induced dipole-induced dipole or van der Waals forces. Such forces are present between various atoms and molecules. These are observed generally in non-polar molecules, halogens, and noble gases.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
- The boiling point of the following compounds is expected to decrease in this order (highest boiling point first).arrow_forwardRank molecules A-D (1=lowest, 4=highest) in order of increasing boiling point. Briefly rationalize your ranking. Please answer all parts.arrow_forwardLabel the kind of intermolecular interaction that will dominate in each of the following molecules.arrow_forward
- I find it pretty hard to understand the difference in boiling points when just given the shape of the molecule. Please help!arrow_forwardCircle all of the following that exhibit ONLY dispersion attractions between molecules. Explain your choices. A) XeF4 B)AsH3 C)CO2 D) CF4 E)H2 F)CHCl3arrow_forwardSelect the diatomic molecule below that has the highest boiling point. * F₂ Cl₂ Br₂ I₂arrow_forward
- Explain the difference between the nature of the bonds between water molecules and those found between ethyl alcohol molecules, and how that plays a role in what you observed.arrow_forwardOrder the molecules by increasing low to high boiling pointsarrow_forwardWhich of these molecules exhibit hydrogen bonding? H. H. H -C-H H-C-CI H C -OH H. H. H. methane chloromethane methanol Select all that apply. methanol chloromethane methanearrow_forward
- Which is neither polar nor capable of hydrogen bonding? Explain with example?arrow_forwardWhich of the following sets of atoms will NOT form a strong hydrogen bond? (Explain your answer) a. C-H .. 0-C b. 0-H .. N ....... c. N-H d. 0-H ....... .......arrow_forwardThe hydrides of group 5A are NH3, PH3, ASH3, and SBH3. Arrange them from highest to lowest boiling point. Rank the molecules from highest to lowest boiling point. To rank items as equivalent, overlap them. • View Available Hint(s) Reset Help SbH, Аянз PH3 NH3 Highest Lowest The correct ranking cannot be determined.arrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

