
Concept explainers
(a)
Interpretation:
The most acidic hydrogen should be circled in the following molecule and the amount of energy which is required to remove the most acidic hydrogen should be calculated.
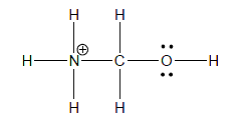
Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A
The value that defines the relationship between the amount of products and reactants at equilibrium is known as equilibrium constant.
(b)
Interpretation:
The expected value of
Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A chemical reaction which takes place between an acid and a base is known as acid-base reaction.
The value that defines the relationship between the amount of products and reactants at equilibrium is known as equilibrium constant.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
- What is the conjugate base of HSO4? Select one: O a. SO 2- O b. OH- O c. H2SO4 O d. HSO+ O e. H,SO,+arrow_forward3. Which of the following species is the strongest base? Multiple Choice HO H2N CH3CO0 CIarrow_forward3. Identify the more acidic compound AND explain why. Draw the conjugate base of each compound. If your answer is "resonance", draw at least 1 resonance structure. H₂S a.) Hz Ot + b.) OH SH US US US GH CH 8arrow_forward
- Rank the structures from most acidic (1) to least acidic (3). H-Br A A B H-CI B [Choose ] [Choose ] [Choose ] > H-I сarrow_forward5. Rank the highlighted protons from most acidic to least acidic. А. Ha ÓH most acidic least acidic OHd на Hb most acidic least acidic B.arrow_forwardQuestion 13 v FLAG QUESTION 1. Dissolving NaNH, in water will give: Answers A - D A. A solution containing solvated Na+ and NH -2 ions. В A. A solution containing solvated Na+ ions, HO ions, and NH,. C A. NH, and metallic Na. D A. Solvated Na ions and hydrogen gas.arrow_forward
- 11. Circle the most acidic protons in the molecules, A and B (one for each molecule). Which compound is more acidic? Why? B H. H. H. H. H H H H.arrow_forwardSelect the strongest base. Đ NH2 ⒸOH OCH3CH₂ O CH2-CH Prev o new datarrow_forwardWhat is/are the most acidic proton() in the moloecle below? Select the anneer where the most acidic proton(s) are circled 0.00 C A H₂ CH₂ che de ch (H₂) D CH₂arrow_forward
- Circle the more acidic compound of each pair. HO но, HO, HO, OH HO, HO, HO, Meo HO. HO, HO. HO. O2Narrow_forwardWhat is the conjugate base of HSO4- is ???? a. SO4 2- b. H2SO4 c. H3O+ d. None correctarrow_forwardWhich of the following is the strongest base? O a. Cl-CH2CH2NH2 Ob. Cl-CH2CH2CH2NH2 O c. Cl-aromatic ring-NH2 O d. Cl-CH2 -NH2 O e. C12-aromatic ring -NH2arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
