
Concept explainers
Interpretation: Each strong acid and strong base in below table should be identified.
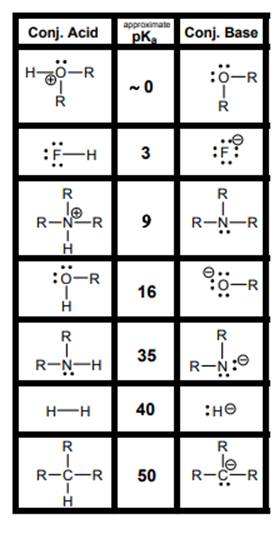
Concept introduction: According to the Bronsted-Lowry concept, substance that donates proton is termed as acid while that accepts or gains protons is called base. Species formed after loss of protons from acids are known as their respective conjugate bases whereas conjugate acid is produced by addition of protons to base. The strength of conjugate acids and conjugate bases are inversely related to the strengths of their respective bases and acids.
According to Lewis concept, the substance that donates electron pair is termed as base while that accepts or gains electron pair is called acid. For example,
According to the Arrhenius concept, substances that donate hydrogen ions in solutions are known as acids while bases are substances that release hydroxide ions in solutions.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
- Is bond formation endothermic or exothermic? Write a + or sign above the arrow in the previousquestion to represent the sign of the energy change associated with the arrow.arrow_forwardPlease help with drawing the conjugate acid and filling in the blank. For "A negative, no charge, positive charge" you have to select the right one. 1. Draw the conjugate acid with formal charge(s) below. If there are electrons around any of the atom signore them for now. The details will be addressed in the next section. 2. N with 3 bonds and 2 electrons around the atom in the base becomes a N atom with (BLANK) bonds. (A negative charge, no charge, positive charge) , and (BLANK) electron(s) around the atom in the conjugate acid.arrow_forward#11 - I need help with only the second row. Row 1 and 3 are correect. The answer is not -11 for row 2. Please help.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
