
Concept explainers
(a)
Interpretation:
The acid-base reaction should be described between the following reactant by using curved arrows and each curved arrow having positive or negative should be marked. The value of

Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A
(b)
Interpretation:
The acid-base reaction should be described between the following reactant by using curved arrows and each curved arrow having positive or negative should be marked. The value of

Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A chemical reaction which takes place between an acid and a base is known as acid-base reaction.
(c)
Interpretation:
The acid-base reaction should be described between the following reactant by using curved arrows and each curved arrow having positive or negative should be marked. The value of
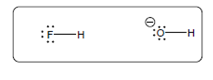
Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A chemical reaction which takes place between an acid and a base is known as acid-base reaction.
(d)
Interpretation:
The acid-base reaction should be described between the following reactant by using curved arrows and each curved arrow having positive or negative should be marked. The value of
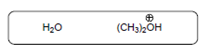
Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A chemical reaction which takes place between an acid and a base is known as acid-base reaction.
(d)
Interpretation:
The acid-base reaction should be described between the following reactant by using curved arrows and each curved arrow having positive or negative should be marked. The value of
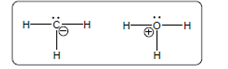
Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A chemical reaction which takes place between an acid and a base is known as acid-base reaction.
(d)
Interpretation:
The acid-base reaction should be described between the following reactant by using curved arrows and each curved arrow having positive or negative should be marked. The value of
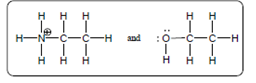
Concept Introduction:
A substance which produces
A substance which produces hydroxide ion when dissolved in aqueous solution is known as a base. These are proton acceptor.
A chemical reaction which takes place between an acid and a base is known as acid-base reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
- Consider the structure of a base. Xyk H Modify the structure to draw the conjugate acid. Add or delete atoms, bonds, charges and non-bonding electrons where appropriate. Draw Rings More Select G |||||| S C H N الا H Erase Q2Qarrow_forwardFor the list of alcohols, rank the alcohols in strength from weakest acid to strongest acid. Weakest Strongest Answer Bank F₂CHOH F, COH FCH₂OH CH₂OHarrow_forwardWhich of the following molecules is the strongest acid? (look at the picture to be able to answer) A B C D None of these are correctarrow_forward
- Which of the compounds shown here do you expect to be the stronger base? Explain. NH NH, orarrow_forwardWhich of these structures has an acidic H?arrow_forwardAde ed arrows to show the Arte attack the proton of the acid (breaking the X-H bond * Identify the nucleophile and electrophile ectants but the charge will be on a different molecule.) Circle the conjugate base. charges of any lons OK a) HO, b) кон SH c) NO. ONa HO + Approximate pK, values of Important organic functional groups pk, Protonated Cacborylic alcohol R. -2 5. 10 Phenol 16 Alcohol 25 Alkyne 38 Amine acid 44 Alkene 50 1. H OH R-OH R -H Alkane R. R. он R H ROH Protonated N amine H. H Thiol R-SHarrow_forward
- Rank the following compounds in decreasing order of acidity. Br A B D OH Br E III > IV>II>I C) III >II> IV>I || > IV > III > II IV > III >II>I IV> || > ||| > | Br = || plan OH Br Br Br OH Br IV OHarrow_forwardSummarize the relationship between pKa and base strength by completing the followingsentences: a. For a given base, the higher the pKa of its conjugate acid, the stronger or weaker the base. b. For a given base, the lower the pKa of its conjugate acid, the stronger or weaker the base.arrow_forwardFor each molecule below, draw the conjugate acid or conjugate base or both if the molecule hasboth a conjugate acid and a conjugate base (e.g., water).arrow_forward
- Answer true or false to the following statements about the mechanism of acid-base reactions. (a) The acid and base must encounter each other by a collision in order for the proton to transfer. (b) All collisions between acids and bases result in proton transfer. (c) During an acid-base reaction the lone pair on the base fills the A-H antibonding sigma orbital.arrow_forwardSeveral acids and their respective equilibrium constants are: Which is the strongest acid? Which is the weakest acid? Which acid has the weakest conjugate base? Which acid has the strongest conjugate base?arrow_forwardThe following are equivalent ways of asking about the acidity of an H atom: • What is the most acidic H on the molecule? • Which H is associated with the published pKa value? • Which H on the molecule is easiest to remove? • Which H on the molecule takes the least energy to remove? • Which bond to an H is most polarized? • For which H atom is removal least uphill in energy? • Which bond to an H atom, when broken, results in the lowest PE conjugate base? We will often find the last of these questions is easiest to answer. To do this, find all the different Hatoms on the molecule, and draw all possible conjugate bases.Only the lowest-energy one is the “real” conjugate base. Identify this structure, and you have found the most acidic H. Use this strategy to find the most acidic H on each of the following molecules. Note: Each structure hasat least three different kinds of H’s, so draw at least three unique conjugate bases for each.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


