
Concept explainers
One method for growing thin silicon sheets for photo voltaic solar panels is to pass two thin strings of highmelting temperature material upward through a bath ofmolten silicon. The silicon solidifies on the strings nearthe surface of the molten pool, and the solid silicon sheet is pulled slowly upward out of the pool. The silicon isreplenished by supplying the molten pool with solid silicon powder. Consider a silicon sheet that is
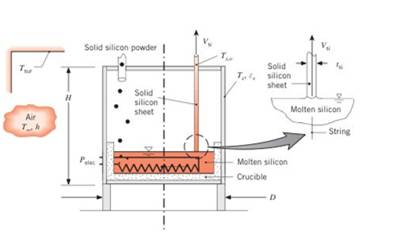
(a) Determine the electric power,
(b) 1f the photovoltaic panel absorbs a time-averagedsolar flux of
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Fundamentals of Heat and Mass Transfer
- What are the diagnostic criteria in Cyclic Voltammetry for a reversible process and irreversible processarrow_forwardTo ensure a tight fit, the aluminum rivet used in airplane construction are made slightly larger than the rivet holes and cooled by dry ice (solid CO2) before being driven. If the diameter of a hole is 0.500 cm, what should be the diameter of a rive at 20 degree Celsius if its diameter is equal that of a hole when the rivet is cooled to -78 degree Celsius, the temperature of dry ice? Assume the coefficient of expansion to remain constant. (Given and solutions)arrow_forwardCalculate the capillary effect in mm in a glass tube of 3mm diameter, when immersed in (1)water, (2)mercury. The temperature of the liquid is 25°C and the values of surface tensions of water and mercury at 25°C in contact with air is 0.0765 and 0.56N/m respectively. The angle of contact for water is 0° and 130° for mercury. Take the density of water 1000 kg/m³ , specific gravity of mercury is 13.6. (ENTER ONLY THE VALUES BY REFERRING THE UNITS GIVEN) The capillary effect of water in mm is equal to=, (1 ma The capillary effect of mercury in mm is equal to=arrow_forward
- Calculate the capillary effect in mm in a glass tube of 4mm diameter, when immersed in (1)water, (2)mercury. The temperature of the liquid is 25°C and the values of surface tensions of water and mercury at 25°C in contact with air is 0.0725 and 0.52N/m respectively. The angle of contact for water is 0° and 130° for mercury. Take the density of water 1000 kg/m3, specific gravity of mercury is 13.6. The capillary effect of water in mm is equal to= The capillary effect of mercury in mm is equal to=arrow_forwardIn boiling water at 1 atm pressure outside a stainless-steel tube with a surface temperature of 410F, the heat-transfer coefficient h in the absence of radiation is 32 Btu/h*ft^2*F. If the emissivity of the stainless steel is 0.8, will radiation significantly augment the rate of boiling (e.g., by more than 5 percent)? Assume that the vapor film is transparent to radiation and the boiling liquid is opaque.arrow_forwardThe hot water coming out of a hydrothermal vent is sometimes called vent fluid. Why would you and I find these vent fluids to be an unpleasant thing to be exposed to? a They are some of the coldest fluids on Earth, being much colder than the normal freezing point of water. b They can be up to 400 degrees Celsius, and contain dissolved compounds like hydrogen sulfide, carbon dioxide, and methane. c The vent fluids are actually an exotic type of magma, made of silicate minerals. d They are actually ultra-pure water, with nothing at all dissolved in them; so pure that even simple cells will die due to the lack of nutrients in the fluids.arrow_forward
- (c) The diameter of the piston and qutled,of the paint sprayer in Fig. 1 is 6 cm and 0.5 cm, respectively. The stroke_of thepston is 25 cm and the height difference between the liquid level in the contairfer'and the upper hole of tube is 12 cm. Calculate the minimum velocity of the piston during pumping to initiate atomizing effect if the liquid density is 1200 kg/m³. Take the atmospheric conditions are 20°C and 95 kPa (air density, Patr = 1.13kg/m³), and the liquid reservoir is open to the atmosphere.arrow_forwardA sprue is 250 mm long. The velocity of the molten metal at the top of the sprue is 0.15 m/s. The middle diameter of the sprue is 10 mm. The total cavity of the "runner system + mold" is 2.5 × 106 mm³. The viscosity of the molten metal is 0.0022 kg/(m · s) and the density is 2700 kg/m³. According to given information i. For a proper sprue design, calculate the bottom diameter of the sprue ii. Find the velocity at the bottom of the sprue iii. Find the volumetric and mass flow rates at the bottom of the sprue iv. Find the required time to fill the mold completely v. Is the flow laminar, mixed or turbulent at the bottom of the sprue? P v? pg' 2g h+-+; = constant Q = A,V1 = A2V2 h2 h1 pVD Re A1 TST = Cm G) A2arrow_forwardOnce upon a time, a student from a certain university wants to learn crystallization using a Swenson-Walker crystallizer. The capacity of this crystallizer is 0.6124 Ib/s of FeSO4 slurry leaves at 300 K. The flow mechanism of the cooling is counter current through the jacket and its temperature increases from 61 to 70 F. The overall heat transfer coefficient has been estimated to be 190 Sl units. (). Determine the requirement for cooling water in kg/hr (i) Let supposed that each crystallizer unit is 3.5 yard long and each metre of crystallizer provides26.91 ft? surface, how many crystallizer units will be required?arrow_forward
- Water is to be boiled at atmospheric pressure in a polished copper pan by means of an electric heater. The diameter of the pan is 0.51 m and is kept at 111 deg C. What is the power required to boil the water? Tsat = 100°C; Properties of water at 100°C: Density, ρl= 961 kg/m3; Kinematic viscosity, ν = 0.293x10-6 m2/s; Prandtl Number, Pr = 1.740; Specific heat, Cpl = 4216 J/kg.K; Dynamic viscosity, μ = ρ × ν = 961 × 0.293 × 10-6 = 281.57 x10-6 Ns/m2; hfg = 2256.9 kJ/kg; ρv = 0.597 kg/m3; σ = 0.0588 N/m; Csf=0.013; n=1; Select one: a. 25964.0356 b. 52997.1101 c. 49373.4011 d. 38911.7986arrow_forwardDraw( insert image) the parts of the Industrial Water Tube Boiler. And discuss the function of each part.arrow_forwardVery pure liquid water can be subcooled at atmospheric pressure to temperatures well below 0 °C. Assume that 1 kg has been cooled as a liquid to -6 °C. A small ice crystal (of negligible mass) is added to “seed” the subcooled liquid. If the subsequent change occurs adiabatically at atmospheric pressure, what fraction of the system freezes and what is the final temperature? What is ΔStotal for the process, and what is its irreversible feature? The latent heat of fusion of water at 0 °C is 333.4 J/g, and the specific heat of subcooled liquid water is 4.226 J/g °C.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





