
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 30.SE, Problem 41AP
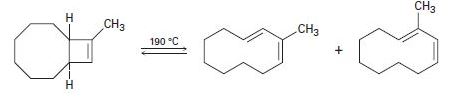
In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Determine the reactants to be used to proceed the following synthesis.
Please help me with reactions 4-6!
Which statement correctly describes the expected major product of the reaction sequence
shown. Assume standard workup as appropriate.
1) BH3, THF
2) НО, Н-О,
3) CrOз, Руridine,
CH,Cl,
O It contains boron
O It has a molecular formula is CęH1002
The product of the second step is the same as the product of the third step, because no reaction occurs in the third step
O It is a primary alcohol
O It is an aldehyde
HO
.OH
cat. H2SO4
H₂O
cat. H2SO4
Complete both mechanisms above. BRIEFLY explain
how the different reaction conditions (in the
presence of acid) result in different products being
formed. How does equilibrium play a role?
Chapter 30 Solutions
Organic Chemistry
Ch. 30.1 - Prob. 1PCh. 30.3 - Prob. 2PCh. 30.3 - Prob. 3PCh. 30.4 - Prob. 4PCh. 30.6 - What stereochemistry would you expect for the...Ch. 30.6 - Prob. 6PCh. 30.7 - Prob. 7PCh. 30.8 - Propose a mechanism to account for the fact that...Ch. 30.8 - When a 2, 6-disubstituted allyl phenyl ether is...Ch. 30.9 - Prob. 10P
Ch. 30.SE - Predict the product obtained when the following...Ch. 30.SE - Prob. 12VCCh. 30.SE - The following rearrangement of N-allyl-N,...Ch. 30.SE - Plastic photochromic sunglasses are based on the...Ch. 30.SE - Prob. 15MPCh. 30.SE - Prob. 16MPCh. 30.SE - Prob. 17MPCh. 30.SE - Prob. 18APCh. 30.SE - Prob. 19APCh. 30.SE - Prob. 20APCh. 30.SE - Prob. 21APCh. 30.SE - Prob. 22APCh. 30.SE - Prob. 23APCh. 30.SE - Prob. 24APCh. 30.SE - Prob. 25APCh. 30.SE - Prob. 26APCh. 30.SE - Prob. 27APCh. 30.SE - Prob. 28APCh. 30.SE - Propose a pericyclic mechanism to account for the...Ch. 30.SE - Prob. 30APCh. 30.SE - Prob. 31APCh. 30.SE - Prob. 32APCh. 30.SE - Prob. 33APCh. 30.SE - Bicyclohexadiene, also known as Dewar benzene, is...Ch. 30.SE - Prob. 35APCh. 30.SE - Prob. 36APCh. 30.SE - The 1H NMR spectrum of bullvalene at 100 C...Ch. 30.SE - Prob. 38APCh. 30.SE - Prob. 39APCh. 30.SE - Prob. 40APCh. 30.SE - In light of your answer to Problem 30-40, explain...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete the following reaction schemes:arrow_forwardIdentify the most likely intermediate in the mechanism leading to the product. НО НО НО. НО OH OH OH OH eTextbook and Media НО НО. Но OH OH _ОН НО. OHarrow_forwardH9.26 - Level 1 := Unanswered • 3 attempts left Compound A can be converted into compound E when treated with concentrated H2SO4. This reaction mechanism proceeds via three intermediates (compounds B-D). Sort the intermediates in the correct order according to the reaction mechanism. Compound C Compound B Compound D Your answer OH compound A compound E intermediates OH2 B Darrow_forward
- NH2 multiple steps OMe OMe 7. Fill in the boxes for each of the following reaction sequences. кон NH DMF solvent „NH2 OHYH,O side product main product 1) LIAIH, Br 2) H,O* NH2arrow_forward9 Complete the following reactions by briefly explaining in each case the type of reaction taking place. allo iii. iv. NOH TT. H₂SO4 A H₂O ??? ??? + ???arrow_forward6. Predict the reactant (starting material) for the reaction and propose a mechanism. NaCN SN2 NaOH E2 CN a Ph Pharrow_forward
- 2) For the following reactions, with arrow pushing shown, please indicate whether the reaction/products/arrow pushing is possible (as drawn with curved arrows). If its not possible, briefly describe what the error is. H2N H4N NH4 + OH H3Narrow_forwardSelect the correct product for this reaction Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. a b C d Compound A Compound B Compound C Compound D 8-686arrow_forwardPlease complete reactions in clear handwritten of all subpartsarrow_forwardChemistry g) Predict starting materials G - J structure for Reaction 5 and 6. Me OTMS D Ph si GH Me OTMS TiCl₂ CH₂Cl₂, -78 °C TiCl₂ CH₂Cl₂, -40 to 0°C OTMS E H Reaction 3 OMe TIC14 H* F O O OH flation N Me OH Reaction 4 Reaction 5 Reaction 6arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
SAR of Anticancer(Antineoplastic) Drug/ Alkylating agents/ Nitrogen Mustard; Author: Pharmacy Lectures;https://www.youtube.com/watch?v=zrzyK3LhUXs;License: Standard YouTube License, CC-BY