
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 30.SE, Problem 34AP
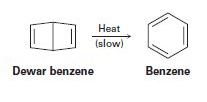
Bicyclohexadiene, also known as Dewar benzene, is extremely stable despite the fact that its rearrangement to benzene is energetically favored. Explain why the rearrangement is so slow.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
In free-radical substitution reaction of alkanes with halogens under uv light,
the photolytic breaking of the halogen is the rate determining step.
the formation of alkylradical is the rate determining step.
the formation of halogen radical is the rate determining step.
the abstraction of hydrogen from alkane by the halogen radical is the rate determining step
The reaction of 3-methylene-1-cyclohexene and HBr yields the four products shown in the attachment. Which two are formed at high temperatures and which two are formed at low temperatures? Why? Why is 1-bromo-3-methylenecyclohexane not formed?
Dehydrohalogenation of 1-chloro-1-methylcyclopropane affords two alkenes (A and B) as products. Explain why A is the major product despite the fact that it contains the less substituted double bond.
Chapter 30 Solutions
Organic Chemistry
Ch. 30.1 - Prob. 1PCh. 30.3 - Prob. 2PCh. 30.3 - Prob. 3PCh. 30.4 - Prob. 4PCh. 30.6 - What stereochemistry would you expect for the...Ch. 30.6 - Prob. 6PCh. 30.7 - Prob. 7PCh. 30.8 - Propose a mechanism to account for the fact that...Ch. 30.8 - When a 2, 6-disubstituted allyl phenyl ether is...Ch. 30.9 - Prob. 10P
Ch. 30.SE - Predict the product obtained when the following...Ch. 30.SE - Prob. 12VCCh. 30.SE - The following rearrangement of N-allyl-N,...Ch. 30.SE - Plastic photochromic sunglasses are based on the...Ch. 30.SE - Prob. 15MPCh. 30.SE - Prob. 16MPCh. 30.SE - Prob. 17MPCh. 30.SE - Prob. 18APCh. 30.SE - Prob. 19APCh. 30.SE - Prob. 20APCh. 30.SE - Prob. 21APCh. 30.SE - Prob. 22APCh. 30.SE - Prob. 23APCh. 30.SE - Prob. 24APCh. 30.SE - Prob. 25APCh. 30.SE - Prob. 26APCh. 30.SE - Prob. 27APCh. 30.SE - Prob. 28APCh. 30.SE - Propose a pericyclic mechanism to account for the...Ch. 30.SE - Prob. 30APCh. 30.SE - Prob. 31APCh. 30.SE - Prob. 32APCh. 30.SE - Prob. 33APCh. 30.SE - Bicyclohexadiene, also known as Dewar benzene, is...Ch. 30.SE - Prob. 35APCh. 30.SE - Prob. 36APCh. 30.SE - The 1H NMR spectrum of bullvalene at 100 C...Ch. 30.SE - Prob. 38APCh. 30.SE - Prob. 39APCh. 30.SE - Prob. 40APCh. 30.SE - In light of your answer to Problem 30-40, explain...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A novel heterocyclic compound, M, contains nitrogen and sulfur atoms and exhibits interesting photochemical properties. When exposed to ultraviolet (UV) light at a specific wavelength, M undergoes a photoreaction resulting in two products: N and O. Product N is formed through a [2+2] cycloaddition involving the nitrogen atom in M, while product O results from a homolytic cleavage of a sulfur-sulfur bond in M. Given these reaction pathways, what is the most probable structure of M, and what are the likely structures of N and O? A. M is a thiazole derivative; N is a dimerized product through the nitrogen atoms, and O is a compound with two sulfur-centered radicals. B. M is a diazine derivative; N is a tetra-atomic cyclic compound, and O is a compound with two separate thiol groups. C. M is a thiadiazole derivative; N is a four-membered ring involving the nitrogen atom, and O results in two sulfur-centered radicals. D. M is a dithiolane derivative; N is a dimer involving the nitrogen…arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + + H₂O **** H₂S04 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH3 CHCCH3 | | OH CH3 +1arrow_forwardfluorination of alkanes is highly exothermic. Per Hammond’s postulate, assume that the transition state for radical fluorination is almost identical to the starting material. Assuming this fact, estimate the fraction of each monofluoro product formed in the fluorination of 2-methylbutane.arrow_forward
- When bromine adds to the double bond in a cyclohexene, the resulting dibromide is trans. This stereochemistry is due to: a) the intermediate bromonium ion b) the intermediate carbocation c) Markovnikov’s rule d) Zaitsev’s rulearrow_forward4. A double bond in a six-membered ring is usually more stable in an endocyclic position than in an exocyclic position. Hydrogenation data on two pairs of compounds follow. One pair suggests that the energy difference between endocyclic and exocyclic double bonds is about 9 kJ/mol. The other pair suggests an energy difference of about 5 kJ/mol. Which number do you trust as being more representative of the actual energy difference? Explain your answer. endocyclic exоcyclic 107 116 105 110 heats of hydrogenation (kJ/mol)arrow_forwardChlordane, like DDT, is an alkyl halide that was used as an insecticide for crops such as corn and citrus and for lawns. In 1983, it was banned for all uses except against termites, and in 1988, it was banned for use against termites as well. Chlordane can be synthesized from two reactants in one step. One of the reactants is hexachlorocyclopentadiene. What is the other reactant?arrow_forward
- The bicyclic alkene P can be prepared by thermal electrocyclic ring closure from cyclodecadiene Q or by photochemical electrocyclic ring closure from cyclodecadiene R. Draw the structures of Q and R, and indicate the stereochemistry of the process by which each reaction occurs.arrow_forwardExplain why heats of hydrogenation cannot be used to determine the relative stability of 2-methylpent-2-ene and 3-methylpent-1-ene. A, B, and C, each subjected to hydrogenation. The number of rings and π bonds refers to the reactant (A, B, or C) prior to hydrogenation.arrow_forwardOrganic Chemistry Indicate which carbocation is the most stable carbocation and which carbocation is the least stable carbocation?Çok Satırlı Metin.arrow_forward
- Define Electrophilic addition and the Hammond postulate ?arrow_forwardBromine adds to cis- and trans-2-butene to give different diastereomers of 2,3-dibromobutane. What does this say about the mode of addition of bromine to thisalkene?arrow_forwardWhich of the following statements about cycloaddition reactions is not true? Cycloaddition reactions form a cyclic product with two new bonds. The course of the reaction is determined by the symmetry of the molecular orbitals of the products. Cycloaddition reactions are concerted. Cycloaddition reactions are stereospecific.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY