
Concept explainers
Interpretation:
The orbital interaction that represents hyperconjugation in
Concept introduction:
Relative stabilities of carbocations are explained by hyperconjugation. In the case of primary carbocations like
Answer to Problem D.1P
The orbital interaction that represents hyperconjugation in
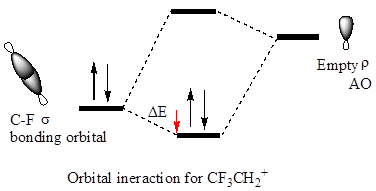
The hyperconjugation leads to greater stabilization for the
Explanation of Solution
In
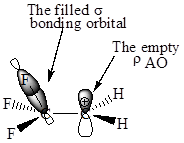
Also, in
The orbital interactions for both

The F atom is more electronegative than H, therefore the
The cation in which hyperconjugation would leads to greater stabilization is determined on the basis of orbital interactions shown above.
Want to see more full solutions like this?
Chapter D Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Two useful organic compounds that contain Cl atoms are vinyl chloride (CH2 = CHCl) and chloroethane (CH3CH2Cl). Vinyl chloride is the starting material used to prepare poly(vinyl chloride), a plastic used in insulation, pipes, and bottles. Chloroethane (ethyl chloride) is a tica roman local anesthetic. Why is the C–Cl bond of vinyl chloride stronger than the C–Cl bond in chloroethane ?arrow_forward2a) The molecule ethene (or ethylene), which has the molecular formula C₂H4, contains two carbon atoms with planar geometry. Construct a model of ethene by first connecting two black balls with two springs. Use four short sticks and four yellow balls to complete the structure. Sketch a perspective representation (a three dimensional drawing) of the structure. 2b) What is the C=C-H bond angle in ethene?arrow_forwardDraw the molecular orbital (MO) electron diagram for the O₂ molecule. Be sure your diagram contains all of the electrons in the molecule, including any core electrons. Energy 1 | MAY X ♫A កarrow_forward
- Sketch the anti bond orbital for the c-cl bond in ch3clarrow_forward1. What are the VSEPR shapes (i.e., molecular geometries) associated with the nitrogen atoms in this molecule? (A) (B) (C) (D) I bent bent bent tetrahedral 2. How many sp² hybridized carbon atoms are present in this structure? (A) 1 HO. Br II trigonal pyramidal N. bent trigonal pyramidal I bent (A) aldehyde, ether, sulfide (B) aldehyde, ketone, thiol (C) amide, ketone, sulfide (D) amide, ether, ester II NH₂ III III trigonal pyramidal (B) 4 (C) 5 (D) 6 3. Which functional groups are NOT present in this molecule? trigonal pyramidal tetrahedral trigonal pyramidalarrow_forward2. In measurements of the structure of molecules, some interesting trends are observed. The bond angle (H-C-H) in CH4 is 109°, as we would expect for a molecule in the tetrahedral VSEPR class. However, other molecules in the same VSEPR class, such as ammonia (NH3, H-N-H= 107°) and water (H2O, H-O-H = 105°), have smaller bond angles than expected. Explain the basis for the bond angle trend: CH4 > NH3 > H2O. %3Darrow_forward
- Determine the angle between covalent bonds in an (SiO4)4- tetrahedron. HINT: Draw a cube, put one O2- anion at the position of 000, another centered on the point with indices of 110, a third at 101, and the fourth at 011. Add one Si4+ cation to the center of the cell. Starting at 000, draw a vector through the center of Si4+. What are the indices of this vector? Starting at 010, draw a second vector through the center towards the O2- at position 101. What are the indices of this vector? Use the (cosφ) equation from Schmid’s law to calculate the angle between the two vectors that you identified.arrow_forwardDraw the Lewis structures and predict the hybridization (sp, sp2, sp3), geometry (linear, trigonal, tetrahedral), and bond angles (180°, 120°, 109.5°) for the central atoms in the following compounds. C and N in C CH3CNarrow_forwardDraw the molecular orbital (MO) electron diagram for the B₂ molecular ion. Be sure your diagram contains all of the electrons in the ion, including any core electrons. Energy X 1 | Sarrow_forward
- CTQ 7: Why is it incorrect to describe the process of boiling as 2H,0(1) → 2H,(g) + 0,(g)?arrow_forwardWhich statement about the CH2=NH molecule is CORRECT? (1) The size of the H-C-H bond angle is 130° (2) the molecule has a trigonal planar structure (3) The pi (TT) bond is formed by the overlap of ONE p-atomic orbital of carbon with ONE s atomic orbital of nitrogen (4) The carbon atom is sp3 hybidizedarrow_forwardWhich compound has a larger dipole moment: CH3Cl or CH2Cl2?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

