
(a)
Interpretation:
The member that rapidly undergoes
Concept Introduction:
The
The first step of
Frist step is the slow step also rate determining step so the rate of the reaction is depends on the concentration of substrate only.
Nucleophile attacks the both front and back side of carbocation in
Order of the substrate that favored in
(a)
Answer to Problem 9.25P
The member that rapidly undergoes
Explanation of Solution
Given:

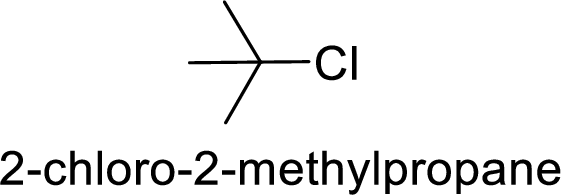
In
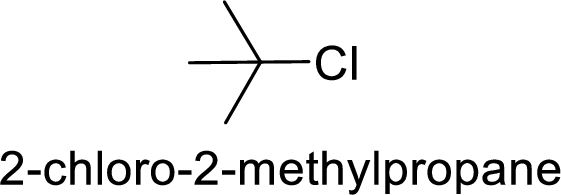
2-chloro-2 methyl propane gives more stable carbocation than 1-chlorobutane.
Hence, the member that rapidly undergoes
(b)
Interpretation:
The member that rapidly undergoes
Concept Introduction:
The rate of the reaction is depends on a single reactant in reaction is known as
The first step of
Frist step is the slow step also rate determining step so the rate of the reaction is depends on the concentration of substrate only.
Nucleophile attacks the both front and back side of carbocation in
Order of the substrate that favored in
(b)
Answer to Problem 9.25P
The member that rapidly undergoes
Explanation of Solution
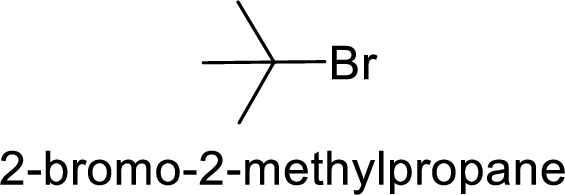
Given:
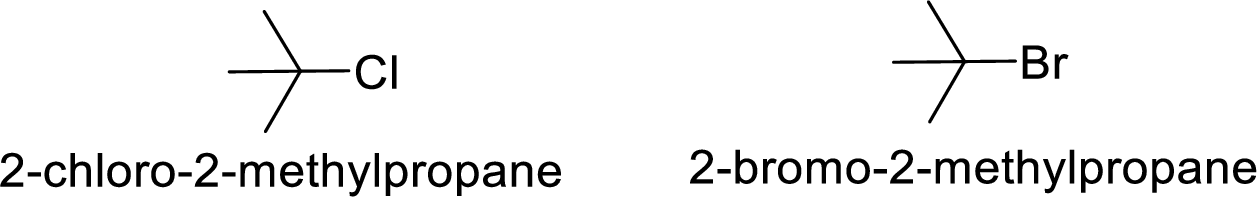
Bromine is better leaving group than chlorine.
Hence, the member that rapidly undergoes
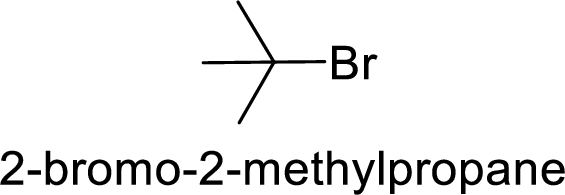
(c)
Interpretation:
The member that rapidly undergoes
Concept Introduction:
The rate of the reaction is depends on a single reactant in reaction is known as
The first step of
Frist step is the slow step also rate determining step so the rate of the reaction is depends on the concentration of substrate only.
Nucleophile attacks the both front and back side of carbocation in
Order of the substrate that favored in
(c)
Answer to Problem 9.25P
The member that rapidly undergoes
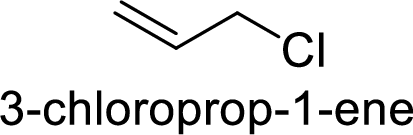
Explanation of Solution
Given:

In
Allyl cation is more stable than primary cation.
Hence, the member that rapidly undergoes
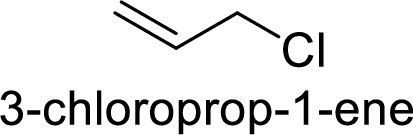
(d)
Interpretation:
The member that rapidly undergoes
Concept Introduction:
The rate of the reaction is depends on a single reactant in reaction is known as
The first step of
Frist step is the slow step also rate determining step so the rate of the reaction is depends on the concentration of substrate only.
Nucleophile attacks the both front and back side of carbocation in
Order of the substrate that favored in
(d)
Answer to Problem 9.25P
The member that rapidly undergoes
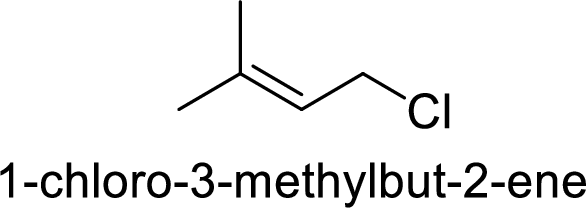
Explanation of Solution
Given:

In
Substuted allyllic cation is more stable than allylic cation.
Hence, the member that rapidly undergoes
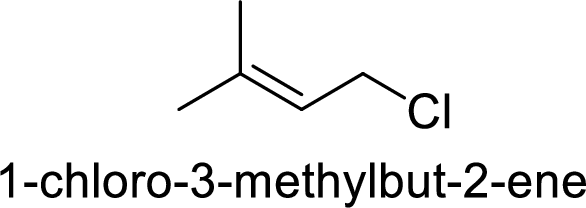
(e)
Interpretation:
The member that rapidly undergoes
Concept Introduction:
The rate of the reaction is depends on a single reactant in reaction is known as
The first step of
Frist step is the slow step also rate determining step so the rate of the reaction is depends on the concentration of substrate only.
Nucleophile attacks the both front and back side of carbocation in
Order of the substrate that favored in
(e)
Answer to Problem 9.25P
The member that rapidly undergoes
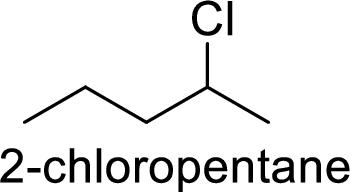
Explanation of Solution
Given:

In
2-chloropentane gives more stable carbocation than 1-chloropentane.
Hence, the member that rapidly undergoes
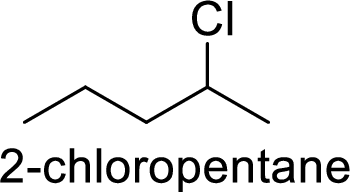
(f)
Interpretation:
The member that rapidly undergoes
Concept Introduction:
The rate of the reaction is depends on a single reactant in reaction is known as
The first step of
Frist step is the slow step also rate determining step so the rate of the reaction is depends on the concentration of substrate only.
Nucleophile attacks the both front and back side of carbocation in
Order of the substrate that favored in
(f)
Answer to Problem 9.25P
The member that rapidly undergoes
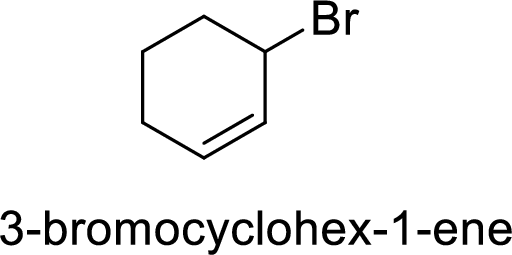
Explanation of Solution
Given:
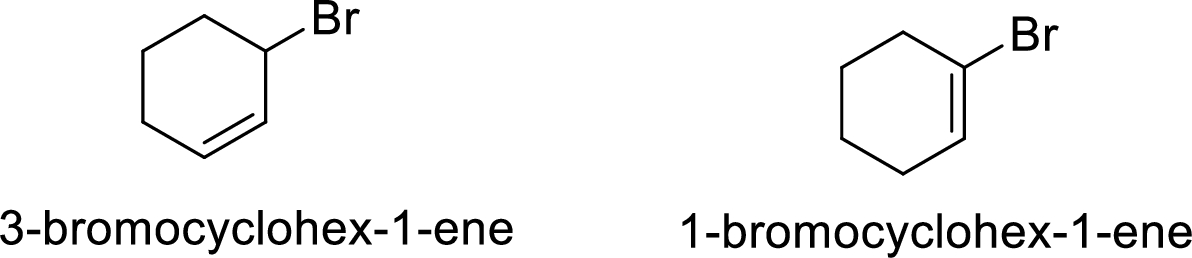
In
Allylic cation is more stable than vinyl cation.
Hence, the member that rapidly undergoes
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry
- Which of the following undergoes SN1 reaction at the fastest rate? Explain.arrow_forwardb) Explain in detail what characteristics of the alkyl halide influence whether a mechanism will be SN1 or SN2. c) Explain in detail what characteristics of a nucleophile influence whether a reaction will be SN1 or SN2.arrow_forwardRank A, B, and C in order of increasing SN1 reactivity.arrow_forward
- Which one of the molecules shown below prefers to exist as its enol tautomer?arrow_forwardwhat is the major product of the SN1 OR SN2 structure below?arrow_forwardWhen bromomethane undergoes solvolysis in a solvent mixture composed of 90% water/10% acetone, the reaction rate is faster than when the same compound is solvolyzed in 80% water/20% acetone. Explain.arrow_forward
- By which substitution mechanism (SN1 or SN2) did the reaction occur? How did you know? For what reason does the substitution occur at only one of the bromine atoms, and in particular the bromine atom that it did?arrow_forwardGive the reaction conditions which could be used to bring about the transformation shown in two stepsarrow_forwardFor each pair of compounds, a state which compound is the better SN2 substrate. (a) cyclohexyl bromide or 1-bromo-1-methylcyclohexanearrow_forward
- i need help filling out the following SN2 reactions with appopiate reactants, products, or reagents,arrow_forwardWhich alkyl halide in each pair will undergo the faster SN2 reaction?arrow_forwardWhich of the following compounds (A or M) would be the better choice for a nucleophilic substitution reactionusing Compound T as the starting material and DMSO as the solvent? Would the resulting reaction be SN1 or SN2?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





