
Interpretation:
The purposeof the use of concentrated
Concept Introduction:
The preparation of aspirin takes place by the reaction of salicylic acid and acetic anhydride represented as follows:
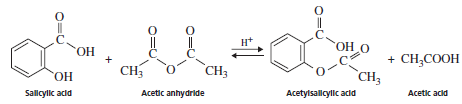
The -OH group of the salicylic acid reacts with acetic anhydride to form ester thus, the formation of aspirin is an esterification reaction.
Answer to Problem 1Q
To activate the reaction.
Explanation of Solution
The acid serves to protonate one double bonded oxygen on the acetic anhydride this makes the reaction with the salicylic acid faster.
The mechanism is represented as follows:
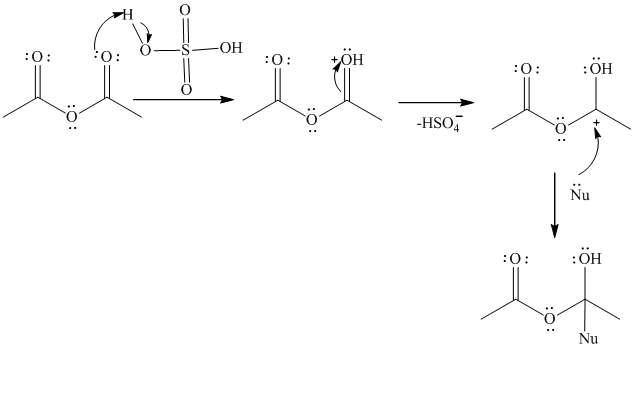
Thus, the acid acts as the catalyst during the esterification reaction by making the carbonyl group of anhydrous acetic acid more reactive.
Thus, the purpose of the use of concentrated
Want to see more full solutions like this?
Chapter 8 Solutions
EBK A SMALL SCALE APPROACH TO ORGANIC L
- The following sequence of reactions occurs in the commercial production of aqueous nitric acid: 4NH3(g)+5O2(g)4NO(g)+6H2O(l)H=907kJ2NO(g)+O2(g)2NO2(g)H=113kJ3NO2+H2O(l)2HNO3(aq)+NO(g)H=139kJarrow_forwardHow can a reaction in an aqueous solution be sped up? A. cooling the reaction mixture B. removing a catalyst C. adding more powdered reactants D. adding excess cold waterarrow_forwardwhy is it necessary to conduct the reaction under acidic conditions?arrow_forward
- Explain the application of coagulation and flocculation process in POME treatment.arrow_forwardPhosphoric and acetic acids are considered to be weak acids; that is, they do not dissociate completely into ions. Were there any differences in reaction rates with magnesium between these two acids? If so, what distinguishing feature of the acids might explain the differences?arrow_forwardWhat are the two reasons why sulfuric acid is added to a filtered solution produced after the aluminum has reacted with the potassium hydroxide?arrow_forward
- What will be the products if the following compounds were to react with ozone and hydrogen peroxide at the same time?arrow_forwardWhat difference(s) do you observe in the cups with and without baking soda as a carbon source? How does the detergent help the leaf disks to sink? What is the purpose of the baking soda solution? Why do the leaf disks in the baking soda solution (treatment) begin to float? Why don't the leaves in the baking soda solution continue to produce oxygen in the dark? If the light-independent reaction can run without light, why does oxygen production (and presumably glucose production) stop? How long did it take for half of the leaf disks to float in each cup?arrow_forwardWhat is wrong with this reaction ?arrow_forward
- Analyze the Sn2 reactions by filling out the chart and answering the questionsarrow_forward6. What would happen to the equilibrium expressed by the reaction in Equation 11.8 if a reagent were added that lowered the Fe3+ ion concentration. a. Equilibrium would shift left. b. Equilibrium would shift right. c. Equilibrium would not change. Explain your answer:arrow_forwardWhat is the Born-Haber process and how is it important to the world’s economyarrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





