
Interpretation:
Whether the conversion of A to B is an exothermic reaction or endothermic reaction should be identified on increase in equilibrium concentration of B.
Concept Introduction:
A chemical equilibrium shifted towards forward direction or backward direction depends on the concentration of both reactant and products.
Exothermic reaction:
A
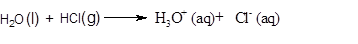
Endothermic reaction:
A chemical reaction in which energy is added when reactant converted into products known as endothermic reaction.
Temperature effect:
In a reaction, reactant molecule collides with each other and many bonds are break and formed in this process; all this process depend on the kinetic energy of molecules and as we increase the temperature the kinetic energy also increases as a result
Consider and equilibrium reaction:
Here, a, b, c and d are coefficient.
If we add excess concentration of c and d, then the chemical equilibrium will shifted in backward direction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Introduction to General, Organic and Biochemistry
- Problem 7-7 Consider the following equilibrium reaction for the decomposition of an aqueous solution of hydrogen peroxide: Oxygen has limited solubility in water (see the table in Chemical Connections 6A). What happens to the equilibrium after the solution becomes saturated with oxygen?arrow_forwardQUESTION 6 The equilibrium constant (K) for an endothermic reaction at 25 deg C is 1.4x10-4. If the temperature increased to 165 deg C what will happen to the value of K? Will be greater than 1.4x10-4 Will be less than 1.4x10-4 Will be equal to 1.4x10 (no change) Not enough information is provided. O O O Oarrow_forwardcomuarrow_forwardProblem 5A is about figuring out the direction of the reaction given initial concentrations. Problem 5B asks what the concentrations will be at equilibrium. Again, this is more difficult and you will need to come back to this part after we cover the necessary material 5A) Suppose the following reaction starts with 1.00 M concentration of HBr, H2, and Br2. If Kc = 1.6 x 10-2, which way will the reaction go? 2 HBr(g) H2 (g) + Br2 (g) 5B) What are the equilibrium concentrations of all the reactants and products?arrow_forwardHeating a metal carbonate leads to decomposition. BaCO3(s) BaO(s) + CO2(g) Predict the effect on the equilibrium of each change listed below. Answer by choosing (i) no change, (ii) shifts left or (iii) shifts right. (a) add BaCO3 (b) add CO2 (c) add BaO (d) raise the temperature (e) increase the volume of the flask containing the reactionarrow_forward7-41 The equilibrium constant at 1127°C for the following endothermic reaction is 571: If the mixture is at equilibrium, what happens to K if we: (a) Add some H2S? (b) Add some H2? (c) Lower the temperature to 1000°C?arrow_forwardEquilibrium reaction Keq expression Stress applied Effect Equilibrium Shi (encircle your answer) (encircle your answer) (increase, decrease, change) in H2le) + Cl2te) 2 HCI(g) Decreasing the Forward pressure no Backward (HCI)2 the No Shift K= ( H2)(Cl2) number of moles of HCl(g) e (increase, decrease, change) in Fe;O4(s) + 4 H2(g) + heat 3 Fe(s) + 4 Increasing Forward temperature Backward no H,O(g) (H20)* the No Shift K= (H2)4 volume of water 2 NO(g) + O,(g) 2 NO2(g) + heat vapor collected f (increase, Addition Forward helium decrease, Backward no K= (NO2)? change) in the No Shift (NO)2 (02) number of moles of NO H,CO3(aq) CO.(aq) + H,0(1) Removal of C (increase, decrease, change) amount of H,CO3 (increase, decrease, change) amount of O2 Forward (CO2) K= no Backward (H2CO3) in the No Shift 4 NH3(g) + 5 0,(g)4 NO(g) H,O(g) + heat Increasing Forward + 6 (NO )* (H20) temperature K= no Backward (NH3) (02)5 in the No Shiftarrow_forwardA reaction occurs according to the equation 2A = Y + 2 Z If in a volume of 5 dm3 we start with 4 mol of pure A and find that 1 mol of A remains at equilibrium, what is the equilibrium constant Kc? Answer: [2.7 mol dm-3 ]arrow_forwardThe reaction C,H2 (g) + 2F2 (9) C,H,F(g) is exothermic in the forward direction. Will an increase in temperature shift the position of the equilibrium toward the reactants or products? O It will shift the position of the equilibrium toward the products. O It will shift the position of the equilibrium toward the reactants. O There is no change on the position of equilibrium. Submit Answer Try Another Version 1 item attempt remainingarrow_forwardissue 6Choose the equilibrium in which the products are favored by a drop in pressure and the reactants favored by a drop in temperature.a) N2(g) + 3 H2(g) ⇔ 2 NH3(g) + 92.3 kJb) H2(g) + I2(g) + 51.8 kJ ⇔ 2 HI(g)c) PCl3(g) + Cl2(g) ⇔ PCl5(g) ΔH = -84.2 kJd) 2 NO2(g) ⇔ 2 NO(g) + O2(g) ΔH = +54 kJarrow_forward◄ Search 5GE 5:20 PM Question 4 of 6 Kc 1000 11% Consider the equilibrium system described by the chemical reaction below. At equilibrium, a 5.0 L reaction vessel contained a mixture of 1.22 mol CH4, 2.79 mol H₂S, 0.630 mol CS₂, and 1.27 × 10-³ mol H₂ at 420.0 °C. What are the values of Kc and Kp for the reaction at this temperature? Complete Parts 1-2 before submitting your answer. CH₂(g) + 2 H₂S(g) = CS₂(l) + 4 H₂(g) 1 2 Based on the given data, set up the expression for Kc. Each reaction participant must be represented by one tile. Do not combine terms. Once the expression is constructed, solve for Kc. = Submit [1.22] [2.79] [0.630] [0.244] [0.558] [0.126] 2[2.79] [2.79]² 2[0.558] 4[2.54 x 10-41 [2.54 x 10-4]4 4[1.27 x 10-³]| Tap here or pull up for additional resources RESET [1.27 x 10-³] [2.54 x 10-4] [0.558]² [1.27 x 10-³]4arrow_forwardFor the equilibrium system N2044) # 2NO26) what is the equilibrium constant when [N2O4) is 1.5 x 10-3 M and [NO2] is 0.57 M? (B) 2.2 × 102 . (A) 3.8 × 10² (C) 4.6 × 10-3 (D) 2.6 × 10-3arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


