
(a)
Interpretation: The expected product for the reactions is to be drawn and stereochemistry along with preferred substitution mechanistic pathway between
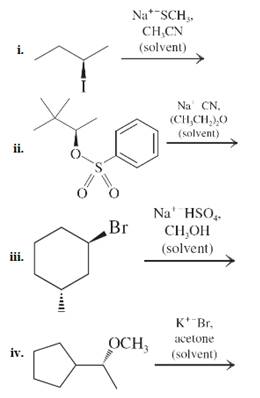
Concept introduction: Unimolecuar substitution or
The general reaction mechanism for the
Step 1: Formation of a carbocation.
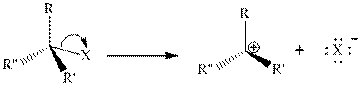
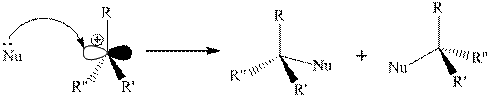
Step 2: Attack of a nucleophile on electron-deficient carbon of a carbocation.
(b)
Interpretation: Whether treatment of
Concept introduction: It has been found experimentally that if the leaving group lies on tertiary carbon than reaction proceeds preferentially via
If the leaving group lies on secondary than both
A good nucleophile can take part in both
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry: Structure and Function
- (b) Consider the reaction of 1-bromobutane with a large excess of ammonia (NH3). Draw the reactants, the transition state, andthe products. Note that the initial product is the salt of an amine (RNH3+ Br - ), which is deprotonated by the excess ammonia to give the amine.arrow_forwardHow will you synthesize cyclohexanecarboxaldehyde (cyclohexylmethanal) from the following reagents? (There are no restrictions on the reagents or the number of steps). (a) Cyclohexanone (b) Ethynylcyclohexane (c) Methyl cyclohexylformate (Remember: Formic acid is the IUPAC recognized name for Methanoic acid) (d) Cyclohexanecarboxylic acid (Cyclohexylmethanoic acid) (e) Vinylcyclohexanearrow_forward(a) Decide whether the reaction below will proceed via an E1 or E2 and write a step-by-step mechanism. (b) Draw structural formula from the major organic product only.arrow_forward
- For each of the substrates below, identify whether: (A) the rate of substitution doesn't depend on nucleophile concentration and (B) the product distribution from substitution gives a 50/50 mix of enantiomers. If you answered "yes" for the first susbtrate, draw the intermediate that forms during a nucleophilic substitution reaction in the space below the table.arrow_forward1-Chlorobutane, CH3CH2CH2CH2Cl, reacts with aqueous sodium hydroxide by a second-order nucleophilic substitution reaction, SN2. a) Draw a diagram to show the mechanism of this reactionarrow_forward(i) State one use each of DDT and iodoform.(ii) Which compound in the following couples will react faster in SN2 displacement and why?(a) 1-bromopentane or 2-bromopentane(b) l-bromo-2-methylbutane or 2-bromo-2-methylbutane.arrow_forward
- 4-Methylpyridine reacts with benzaldehyde (C6H5CHO) in the presence of base to form A. (a) Draw a stepwise mechanism for this reaction. (b) Would you expect 2-methylpyridine or 3-methylpyridine to undergo a similar type ofcondensation reaction? Explain why or why not.arrow_forward4. identify the structure of A and B in the following synthetic scheme: Write out complete reactions for each step, showing the structure of all reactants and products. (a) cyclohexanol +Na2Cr2O7/H2SO4, H2O -------> A (b) A + Et2NH/H2SO4 ----->Barrow_forward1 Consider the reaction of (R)-2-chloro-3-methylbutane with sodium iodide to form aproduct.(a) Draw the reaction scheme with the correct stereochemistry (reactant + NaI → product+ NaCl). Circle the nucleophile and draw a rectangle around the electrophile.(b) What is the symbol used for mechanism shown in 1(a)(c) If the sodium iodide was replaced with sodium hydroxide, the product is anALKENE. Draw a reaction MECHANISM to show how this happens.(d) Draw the reaction energy diagram for the reaction in 1(c) and label the activationenergy. (e) Using any alcohol with five carbons, and any carboxylic acid with six carbons, draw areaction to show how we would make an ester.(f) Describe the practical on esters. Please answer (d) to (f)arrow_forward
- 7. For each pair of compounds, say which compound is the best SN2 substrate.(a) 2-methyl-1-iodopropane or tert-butyl iodide(b) cyclohexyl bromide or 1-bromo-1-methylcyclohexane(c) 2-bromobutane or isopropyl bromide(d) 1-chloro-2,2-dimethylbutane or 2-chlorobutane(e) 1-iodobutane or 2-iodopropanearrow_forwardFor each reaction, give the expected substitution product, and predict whether the mechanism will be predominantly first order (SN1) or second-order (SN2). (a) cyclohexyl bromide + sodium ethoxidearrow_forwardIllustrate the resonance effect of the methoxy group -OCH3, on the structure of the benzene ring. Draw all the oissuvke resonance forms of methoxybenzene, including the hybrid Based on the structures, explain how the presence of the -OCH3 group affects: (i) the reactivity of the benzene ring towards electrophilic attack (ii) the orientation or point of attack of an incoming electrophilic reagent on the benzene ring.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





