
Concept explainers
Interpretation:
The diagrams of waves are to be drawn, the wavelength that X-rays and radio waves have on this scale is to be calculated and the range of wavelength visible to our eyes are to be determined.
Concept introduction:
The distance between two consecutive crests of a wave of light is called the
The arrangement of lights according to the wavelength associated with it, is called a spectrum.
To convert the m to nm, the conversion factor is as:
To convert nm to mm, the conversion factor is as:
Answer to Problem 36E
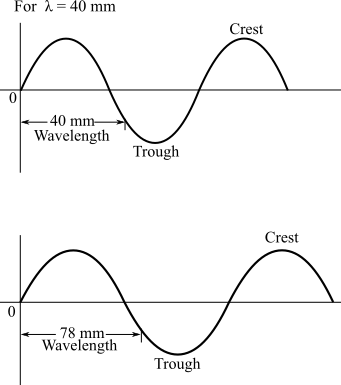
Solution: The diagram of a wave of the violet edge
The wavelength that X-rays and radio waves have on this scale are
Explanation of Solution
Given information:
The violet edge of spectrum appears at a wavelength of
The wavelength of X-rays, radio waves and visible range in mm is calculated as follows:
For X-rays,
For radio waves,
For violet and red edged visible range,
For Violet edge:
For Red edge:
The sketches of the waves associated with wave lengths 40mm and 78 mm are as:

Diagrams of waves are drawn as follows;

The X-rays and radio waves on this scale are
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry In Focus
- Traffic signals are often now made of LEDs (light-emitting diodes). Amber and green ones are pictured here. (a) The light from an amber signal has a wave-length of 595 nm, and that from a green signal has a wavelength of 500 nm. Which has the higher frequency? (b) Calculate the frequency of amber light.arrow_forwardAn FM radio station found at 103.1 on the FM dial broadcasts at a frequency of 1.031188s1 (103.1 MHz). What is the wavelength of these radio waves in meters?arrow_forwarddescribe waves in terms of frequency, wavelength, and amplitude.arrow_forward
- RGB color television and computer displays use cathode ray tubes that produce colors by mixing red, green, and blue light. If we look at the screen with a magnifying glass, we can see individual dots turn on and off as the colors change. Using a spectrum of visible light, determine the approximate wavelength of each of these colors. What is the frequency and energy of a photon of each of these colors?arrow_forwardYou are an engineer designing a switch that works by the photoelectric effect. The metal you wish to use in your device requires 6.7 1019 J/atom to remove an electron. Will the switch work if the light falling on the metal has a wavelength of 540 nm or greater? Why or why not?arrow_forwardThe eyes of certain reptiles pass a single visual signal to the brain when the visual receptors are struck by photons of a wavelength of 850 nm. If a total energy of 3.151014 J is required to trip the signal, what is the minimum number of photons that must strike the receptor?arrow_forward
 Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





