
Concept explainers
Interpretation: The resonance structure of diazomethane should be found.
Concept Introduction: Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance.
In some molecules, there is possibility of more than one Lewis structure where all the structures are equally acceptable. One of the acceptable Lewis structures of these molecules is called resonance structure.
All the possible resonance structures are imaginary whereas the resonance hybrid is real.
Any of the possible structure does not exist as such like a stable real molecule. So it is not possible to isolate one resonance structure.
These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Structure with greater number of covalent bonds are more stable comparing to that with lower number of covalent bonds.
Structure which does not involve charge separation is more stable when comparing with structure having positive and negative charge separation.
While drawing resonance structure of a molecule some rules should be followed where the position, over whole charge and chemical framework remains intact. Also only π and nonbonding electron has been moved in all the three resonance structures
Answer to Problem 6.36QP
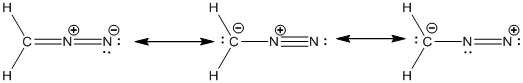
Explanation of Solution
The given atomic arrangement of diazomethane is
Resonance structure of hydrazoic acid is drawn below.

In the case of diazomethane, the chemical bonding of a molecule cannot be represented using a single Lewis structure. The chemical bonding are described by delocalization of electrons forming 3 possible resonance structures. In all the 3 resonance structures the position, over whole charge and chemical framework remains intact.
The resonance structures of the diazomethane molecule were drawn.
Interpretation: The formal charge of diazomethane molecule should be found.
Concept Introduction:
A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all
This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
The Lewis structure with formal charge on each of the atoms close to zero is taken as the most plausible structure.
Formal charge of an atom can be determined by the given formula.
Answer to Problem 6.36QP
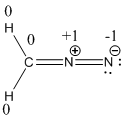
Resonance structure of diazomethane is given below.

Explanation of Solution
The formal charge of the given compound is calculated,
- First hydrogen atom
Substituting these values to the equation,
- Second hydrogen atom
Substituting these values to the equation,
- Carbon atom
Substituting these values to the equation,
- First nitrogen atom,
Substituting these values to the equation,
- Second nitrogen atom
Substituting these values to the equation,
Resonance structure of diazomethane is given below.
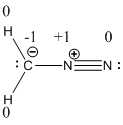
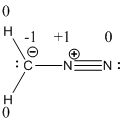
The formal charge of the given compound is calculated,
- First hydrogen atom
Substituting these values to the equation,
- Second hydrogen atom
Substituting these values to the equation,
- Carbon atom
Substituting these values to the equation,
- First nitrogen atom,
Substituting these values to the equation,
- Second nitrogen atom
Substituting these values to the equation,
Resonance structure of the diazomethane is given below.

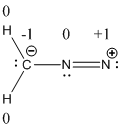
The formal charge of the given compound is calculated,
- First hydrogen atom
Substituting these values to the equation,
- Second hydrogen atom
Substituting these values to the equation,
- Carbon atom
Substituting these values to the equation,
- First nitrogen atom,
Substituting these values to the equation,
- Second nitrogen atom
Substituting these values to the equation,
The formal charges of diazomethane molecule were shown.
Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry: Atoms First
- Write the Lewis structure for nitrosyl fluoride, FNO. Using only a periodic table, identify (a) which is the longer bond. (b) which is the stronger bond. (c) which is the more polar bond.arrow_forwardGiven the bonds C N, C H, C Br, and S O, (a) which atom in each is the more electronegative? (b) which of these bonds is the most polar?arrow_forwardDefine the term lattice energy. Why, energetically, do ionic compounds form? Fig. 3-8 illustrates the energy changes involved in the formation of MgO(s) and NaF(s). Why is the lattice energy of MgO(s) so different from that of NaF(s)? The magnesium oxide is composed of Mg2+ and O2 ions. Energetically, why does Mg2+O2 form and not Mg+O? Why doesnt Mg3+O3 form?arrow_forward
- Bond Enthalpy When atoms of the hypothetical element X are placed together, they rapidly undergo reaction to form the X2 molecule: X(g)+X(g)X2(g) a Would you predict that this reaction is exothermic or endothermic? Explain. b Is the bond enthalpy of X2 a positive or a negative quantity? Why? c Suppose H for the reaction is 500 kJ/mol. Estimate the bond enthalpy of the X2 molecule. d Another hypothetical molecular compound, Y2(g), has a bond enthalpy of 750 kJ/mol, and the molecular compound XY(g) has a bond enthalpy of 1500 kJ/mol. Using bond enthalpy information, calculate H for the following reaction. X2(g)+Y2(g)2XY(g) e Given the following information, as well as the information previously presented, predict whether or not the hypothetical ionic compound AX is likely to form. In this compound, A forms the A+ cation, and X forms the X anion. Be sure to justify your answer. Reaction: A(g)+12X2(g)AX(s)The first ionization energy of A(g) is 400 kJ/mol. The electron affinity of X(g) is 525 kJ/mol. The lattice energy of AX(s) is 100 kJ/mol. f If you predicted that no ionic compound would form from the reaction in Part e, what minimum amount of AX(s) lattice energy might lead to compound formation?arrow_forwardUsing the bond dissociation enthalpies in Table 8.8, estimate the enthalpy of combustion of gaseous methane, CH4, to give water vapor and carbon dioxide gas.arrow_forwardConsider the pyrosulfate ion, S2O72-. It has no sulfur–sulfur nor oxygen–oxygen bonds. (a) Write a Lewis structure for the pyrosulfate ion using only single bonds. (b) What is the formal charge on the sulfur atoms for the Lewis structure you drew in part (a)? (c) Write another Lewis structure using six bonds and two O—S bonds. (d) What is the formal charge on each atom for the structure you drew in part (c)?arrow_forward
- A common trait of simple organic compounds is to have Lewis structures where all atoms have a formal charge of zero. Consider the following incomplete Lewis structure for an organic compound called methyl cyanoacrylate, the main ingredient in Super Glue. Draw a complete Lewis structure for methyl cyanoacrylate in which all atoms have a formal charge of zero.arrow_forwardThe study of carbon-containing compounds and their properties is called organic chemistry. Besides carbon atoms, organic compounds also can contain hydrogen, oxygen, and nitrogen atoms (as well as other types of atoms). A common trait of simple organic compounds is to have Lewis structures where all atoms have a formal charge of zero. Consider the following incomplete Lewis structure for an organic compound called histidine (an amino acid), which is one of the building blocks of proteins found in our bodies: Draw a complete Lewis structure for histidine in which all atoms have a formal charge of zero.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





