
Concept explainers
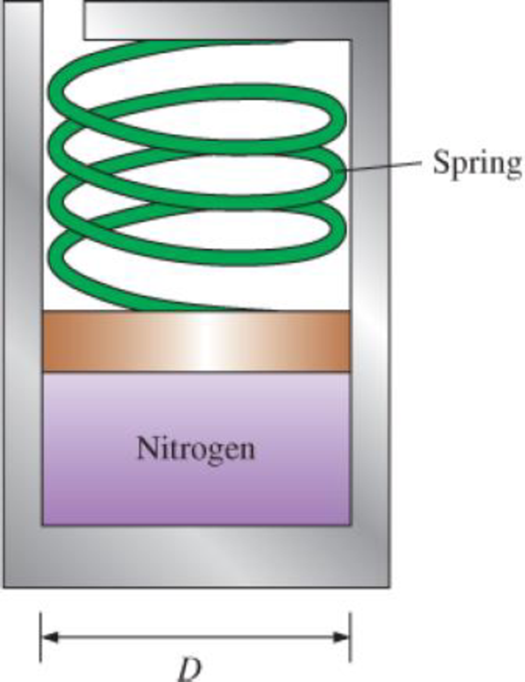
A mass of 10 g of nitrogen is contained in the spring-loaded piston–cylinder device shown in Fig. P4–54. The spring constant is 1 kN/m, and the piston diameter is 10 cm. When the spring exerts no force against the piston, the nitrogen is at 120 kPa and 27°C. The device is now heated until its volume is 10 percent greater than the original volume. Determine the change in the specific internal energy and enthalpy of the nitrogen.
FIGURE P4–54
The change in the internal energy of the nitrogen.
The change in the enthalpy of the nitrogen.
Answer to Problem 54P
The change in the internal energy of the nitrogen is
The change in the enthalpy of the nitrogen is
Explanation of Solution
Write the expression for the initial volume of nitrogen.
Here, the mass of the spring loaded piston cylinder device is
Determine the linear P-v process for spring loaded piston cylinder device.
Here, the system pressure is
Determine the specific heat constant value.
Substitute
From the Equation (IV), the final volume is 10 percent greater than the original volume;
Determine the final temperature of the nitrogen.
Here, the final pressure of the nitrogen is
Determine the internal energy of the spring loaded piston-cylinder device.
Here, the specific heat of constant volume is
Determine the enthalpy of the spring loaded piston-cylinder device.
Here, the specific heat of constant pressure is
Conclusion:
Write the conversion of unit for temperature of 20 C from
Refer Table A-2(a), “Ideal-gas specific heats of various common gases” to obtain the value of gas constant, specific heat of constant volume and pressure for nitrogen gas is
Substitute
Substitute
Substitute
Substitute
Substitute
Thus, the change in the internal energy of the nitrogen is
Substitute
Thus, the change in the enthalpy of the nitrogen is
Want to see more full solutions like this?
Chapter 4 Solutions
Thermodynamics: An Engineering Approach
- A piston–cylinder device contains 0.05 m3 of a gas initially at 200 kPa. At this state, a linear spring that has a spring constant of 150 kN/m is touching the piston but exerting no force on it. Now heat is transferred to the gas, causing the piston to rise and to compress the spring until the volume inside the cylinder doubles. If the crosssectional area of the piston is 0.25 m2 , determine (a) the final pressure inside the cylinder, (b) the total work done by the gas, and (c) the fraction of this work done against the spring to compress it.arrow_forwardWater is contained in a cylinder/piston arrangement fitted with upper stops which limit the volume to 0.2 m maximum. The mass of the water is 0.26 kg, and the weight of the piston and the atmospheric pressure are balanced when the internal pressure is 200kPa. Initially, the piston pushes against the upper stops, and the pressure is 400 kPa. The water is allowed to slowly cool down to a temperature of 110°C. Sketch the process on both the P-v and the T-v diagrams. Label all relevant points and assignarrow_forwardA piston–cylinder device contains 0.85 kg of refrigerant- 134a at 210ºC. The piston that is free to move has a mass of 12 kg and a diameter of 25 cm. The local atmospheric pressure is 88 kPa. Now, heat is transferred to refrigerant-134a until the temperature is 15ºC. Determine: (a) the final pressure,(b) the change in the volume of the cylinder, and (c) the change in the enthalpy of the refrigerant-134a.arrow_forward
- 0.2 m^3 of an ideal gas at a pressure of 2Mpa and 600 k is expanded isothermally to 5 times the initial volume. It is then cooled to 300 K at constant Volume and then compressed back polytropically to its initial state. Show the process on a P-V diagram and determine the work done.arrow_forward100-kg of R-134a at 200 kPa are contained in a pistoncylinder device whose volume is 12.322 m3. The piston is now moved until the volume is one-half its original size. This is done such that the pressure of the R-134a does not change. Determine change in the total internal energy, in kJ/kg, of the R-134a.arrow_forward1-ft³ of air is contained in a spring-loaded piston-cylinder device. The spring constant is 6lbf/in, and the piston diameter is 12 in. When no force is exerted by the spring on the piston, the state of the air is 250 psia and 450°F. This device is now cooled until the volume is one-third its original size. Determine the change in the specific internal energy and enthalpy of the air.arrow_forward
- The spring-loaded piston–cylinder device shown in Fig. P3–63 is filled with 0.5 kg of water vapor that is initially at 4 MPa and 400°C. Initially, the spring exerts no force against the piston. The spring constant in the spring force relation F = kx is k = 0.9 kN/cm and the piston diameter is D = 20 cm. The water now undergoes a process until its volume is one-half of the original volume. Calculate the final temperature and the specific enthalpy of the water.arrow_forward3. A piston-cylinder device contains .85 kg of refrigerant-R134a at -10 °C. The piston that is free to move has a mass of 12 kg and a diameter of 25 cm. The local atmospheric pressure is 88 kPa. Now, heat is transferred to refrigerant-134a until the temperature is 15°C. Determine (a) the final pressure, (b) the change in the volume of the cylinder, and (c) the change in the enthalpy of the refrigerant-134a. R-134a 0.85 kg -10°Carrow_forwardA frictionless piston-cylinder device initially contains 200L of saturated liquid refrigerant-134a. The piston is free to move and its mass is such that it maintains a pressure of 900 kPa on the refrigerant. The refrigerant is now heated until its temperature rises to 70C. Calculate the work done during this process. ANS. 5571 KJA fixed mass of an ideal gas is heated from 50 to 80°C at a constant pressure of (a) 1 atm and (b) 3 atm. For which case do you think the energy required will be greater?arrow_forward
- A 0.8-m3 rigid tank contains nitrogen gas at 600 kPa and 300 K. Now the gas is compressed isothermally to a volume of 0.1 m3. Solve for the work done on the gas during this compression process.arrow_forwardA piston-cylinder device initially contains 300 liters of of water at 430 kPaa and 110 degC. Heat is added to the water at constant pressure until the liquid is entirely vaporize.Determine (a) the amount of heat addedarrow_forwardA mass of 0.1 kg of helium fills a 0.2 m³ rigid vessel at 350 kPa. The vessel is heated until the pressure is 710 kPa. Calculate the temperature change of helium (in K) as a result of this heating. The gas constant of helium is R= 2.0769 kPa-m3/kg-K. Helium 0.1 kg 0.2 m 350 kPa The temperature change is K.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





