
Draw the structure of a compound fitting each description:
a. an
b. a
c. a
d. an ester with molecular formula
(a)
Interpretation: The structure of an aldehyde with molecular formula
Concept introduction: Functional groups are specific substituents present in the molecule that is responsible for the characteristic chemical reactions. For example, functional groups possess a carbonyl group includes aldehydes, ketones, carboxylic acids, amides, esters and acid chlorides. An organic compound containing
Answer to Problem 3.10P
The structure of an aldehyde with molecular formula
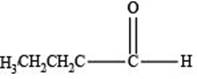
Explanation of Solution
The given molecular formula is
Where,
• C is the number of carbon atoms.
• H is the number of hydrogen atoms.
Substitute the values of number of carbon atoms and hydrogen atoms in the above formula to calculate the number of double bond in compound
Therefore, the given compound has one double bond with an aldehydic functional group. Hence, the structure an aldehyde with molecular formula
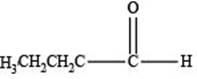
Figure 1
The structure of an aldehyde with molecular formula
(b)
Interpretation: The structure of a ketone with molecular formula
Concept introduction: Functional groups are specific substituents present in the molecule that is responsible for the characteristic chemical reactions. For example, functional groups possess a carbonyl group includes aldehydes, ketones, carboxylic acids, amides, esters and acid chlorides. An organic compound containing
Answer to Problem 3.10P
The structure of a ketone with molecular formula
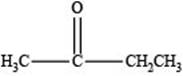
Explanation of Solution
The given molecular formula is
Where,
• C is the number of carbon atoms.
• H is the number of hydrogen atoms.
Substitute the values of number of carbon atoms and hydrogen atoms in the above formula to calculate the number of double bond in compound
Therefore, the given compound has one double bond with a ketone functional group. Hence, the structure a ketone with molecular formula

Figure 2
The structure of a ketone with molecular formula
(c)
Interpretation: The structure of a carboxylic acid with molecular formula
Concept introduction: Functional groups are specific substituents present in the molecule that is responsible for the characteristic chemical reactions. For example, functional groups possess a carbonyl group includes aldehydes, ketones, carboxylic acids, amides, esters and acid chlorides. An organic compound containing
Answer to Problem 3.10P
The structure of a carboxylic acid with molecular formula
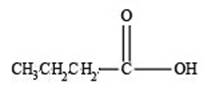
Explanation of Solution
The given molecular formula is
Where,
• C is the number of carbon atoms.
• H is the number of hydrogen atoms.
Substitute the values of number of carbon atoms and hydrogen atoms in the above formula to calculate the number of double bond in compound
Therefore, the given compound has one double bond with a carboxylic acid functional group. Hence, the structure a carboxylic acid with molecular formula
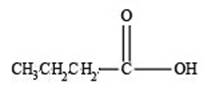
Figure 3
The structure of a carboxylic acid with molecular formula
(d)
Interpretation: The structure of an ester with molecular formula
Concept introduction: Functional groups are specific substituents present in the molecule that is responsible for the characteristic chemical reactions. For example, functional groups possess a carbonyl group includes aldehydes, ketones, carboxylic acids, amides, esters and acid chlorides. An organic compound containing
Answer to Problem 3.10P
The structure of an ester with molecular formula
Explanation of Solution
The given molecular formula is
Where,
• C is the number of carbon atoms.
• H is the number of hydrogen atoms.
Substitute the values of number of carbon atoms and hydrogen atoms in the above formula to calculate the number of double bond in compound
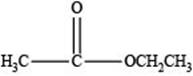
Therefore, the given compound has one double bond with an ester functional group. Hence, the structure an ester with molecular formula

Figure 4
The structure of an ester with molecular formula
Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry
- 1. An amine is characterized by what functional group? a. -CO2CH3 b. -NH2 d. -CHO e. -OH c. -CO2H 2. Which of the follow ing is an akohol? a. NaOH d. CH CH 20OH b. H3CNH2 c. H3C-C-CH3 с. 3. When two alcohols undergo a self condensation, what is formed? a. liquid alcohol b. a ketone d. an aklehyde an ether e. с. an esterarrow_forwardketone a. carbonyl group carboxylic acid b. hydroxyl group 3. alkane c. general formula R3N 4. alkyne d. triple bond 5. ester e. smell of fruits 6. alcohol f. fuels amine g. vinegar 8. hydrocarbon h. carbon and hydrogenarrow_forward24 Which of the following contains a hydroxyl group? A. Alcohol B. Ether C. Thiol D. More than one answer is correct 25 Tetrahydrocannabinol molecule has four functional groups. Which one is not included? A. An alkane B. an ether C. a phenolic -OH D. benzene ring E. a carbon-carbon double bond H H3C CH3 H3C H OH CH3arrow_forward
- 8. Classify each alkene, alkyne., aromatic hydrocarbon, aleohol,ther ketone, carboxylic acidy Of the following organic compound as alkane , aldehyde amide, an hydrideor amine ester a. CHs CH z CH こ C4C4っ b. C. CH3 CHz CH - CH2O d. COCH3 e. CHz (cHz)8 C Hy f. CHz COO CHz CHz 9. cH, CH, CHz-CH h. CH EC - CHz Ctg i: CHz CH,-く4。 J. cH, C4 - と一6H K: CHs CH, NH CH> 1. CH3 - C-C Hz トH2 CH,arrow_forward1. what grouo does the ff organic compound belong? a. ketone b. ether c. cyloalkane d. esther 2. what group does the ff organic compound belong? a. amide b. azo c. nitrile d. amine 3. what is the priority functional group of the ff organic compound? a. carboxyl b. hydroxyl c. carbonyl d. hydroxidearrow_forwardFunctional Group Classes Alcohol Amide Ester • CH₂CH₂OH * CHIO-CH, 7. H₂C1O Amine Ketone Circle each functional group(s) and identify the class to which each of the groups belongs. 9. H₂C Aldehyde Thiol Aromatics Carboxylic Acid 2. CH,SH 6. Alkenes 4. H₂NCH₂CH₂ 10. Ether NM₂ 11. Which of the functional groups in the top list must be on a terminalC in the carbon chain?arrow_forward
- The major compounds used in Fragrance are: a. Ethers. O b. Alcohols. O c. Carboxylic acid. O d. Anhydrides. Which of the following statement is CORRECT? O a. Ethanol and Propenol. O b. Methanol and Ethanol. c. Ethanol and Propanol. O d. Cyclomethanol and Cycloethanol. The first two members of the homologous series of alcohols are: a. Ethanol and Propenol. b. Methanol and Ethanol. O c. Ethanol and Propanol. O d. Cyclomethanol and Cycloethanol.arrow_forward14. PART 3: Draw the structure for compound A.arrow_forward1. What does the ff compound belong to: a. ketone b. alcohol c. aldehyde d. alkane 2. What group does the ff organic compound belong? a. ketone b. carboxylic acid c. aldehyde d. alkyl halide 3. What group does the ff organic compound belong? a. alkene b. ketone c. alcohol d. aldehydearrow_forward
- What are the major products of the reaction of ethyl benzoate with hydrochloric acid and water? a. acetic acid and toluene b. phenylic acid and ethanol C. ethanoic acid and benzene d. benzoic acid and ethanol e. phenylic acid and methanol O a O barrow_forwardIt is a component of nylon 66. a. Phthalic acid b. Lauric acid O c. Benzoic acid d. Adipic acid e. Ethanoic acid Primary amine can be prepared by a. Reacting an alkyl halide with sodium azide. b. Reacting an amide with water under heat. O c. Reacting alkyl halide with NaCN followed by LiAlH4 in ether, then with water. O d. Reacting carboxylic acid with R-NH2 and NaBH4. Which of the following will give a positive reaction with Tollen's reagent? a. All of the above b. Butanone O c. 3-pentanone Od. Pentanal O e. Butanalarrow_forward1. The functional groups present in the following molecules are: a. a carboxylic acid and an ester b. an ether, a ketone and a carboxylic acid c. an ester, a alcohol and a ketone d. an ether, a ketone and an alcohol c. nonc of the above H3C CH3 H3C- -CH2 CH2 .CH2 -CH2 HO, CH2 CH2arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





