
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22.SE, Problem 37AP
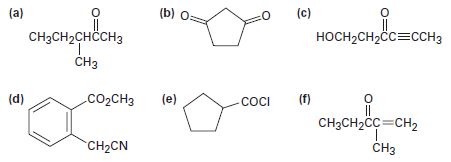
Identify all the acidic hydrogens (pKa < 25) in the following molecules:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Given is teh mass spectogram of an unknown compoound. This unstable cyclic compund is a colorless liquid with a sharp odor and reacts with light and air with long exposure.
What is the molar mass of the target mecule?
What is the Molecular formula of the target molecule?
What IHD /U ?
What could be the description of the structure?
Give the name of the structure.
(30) Take a look at the two multi-substituted benzene compounds below. Trinitrotoluene (TNT) has its NO, groups on carbons 2, 4, and 6.
Which of the two fits this numerical scheme? Hint: Toluene is a special name for benzene whose primary functional group is a methyl group
substituted in place of one of its hydrogens.
CH3
CH3
O,N.
ZON
O,N
NO2
NO2
NO2
e 5 CHM2211
[References]
Carboxylic acids, such as enanthic acid (heptanoic acid, C7H14O₂), often give a strong fragment ion at m/z (M-17). What is the likely structure of this
cation derived from enanthic acid? Show by drawing contributing structures that it is stabilized by resonance.
.
.
.
Only show specific types of hydrogen atoms:
o Hydrogen atoms not attached to carbon atoms
Apply formal charges where appropriate.
Assign lone pairs and radical electrons where appropriate.
Omit arrows and + signs between structures.
✪
**
MELL
///
ChemDoodle
Previous
Next
Chapter 22 Solutions
Organic Chemistry
Ch. 22.1 - Prob. 1PCh. 22.1 - How many acidic hydrogens does each of the...Ch. 22.1 - Prob. 3PCh. 22.3 - Write the complete mechanism for the deuteration...Ch. 22.3 - Prob. 5PCh. 22.4 - If methanol rather than water is added at the end...Ch. 22.5 - Prob. 7PCh. 22.5 - Draw a resonance structure of the acetonitrile...Ch. 22.6 - If methanol rather than water is added at the end...Ch. 22.7 - Prob. 10P
Ch. 22.7 - Draw a resonance structure of the acetonitrile...Ch. 22.7 - Why do you suppose ketone halogenations in acidic...Ch. 22.7 - Prob. 13PCh. 22.7 - Prob. 14PCh. 22.7 - Prob. 15PCh. 22.7 - Prob. 16PCh. 22.SE - Prob. 17VCCh. 22.SE - Prob. 18VCCh. 22.SE - Prob. 19VCCh. 22.SE - Prob. 20MPCh. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Prob. 23MPCh. 22.SE - In the Hell–Volhard–Zelinskii reaction, only a...Ch. 22.SE - Prob. 25MPCh. 22.SE - Nonconjugated , -unsaturated ketones, such as...Ch. 22.SE - Prob. 27MPCh. 22.SE - Using curved arrows, propose a mechanism for the...Ch. 22.SE - Prob. 29MPCh. 22.SE - One of the later steps in glucose biosynthesis is...Ch. 22.SE - The Favorskii reaction involves treatment of an...Ch. 22.SE - Treatment of a cyclic ketone with diazomethane is...Ch. 22.SE - Prob. 33MPCh. 22.SE - Amino acids can be prepared by reaction of alkyl...Ch. 22.SE - Amino acids can also be prepared by a two-step...Ch. 22.SE - Heating carvone with aqueous sulfuric acid...Ch. 22.SE - Identify all the acidic hydrogens (pKa 25) in the...Ch. 22.SE - Rank the following compounds in order of...Ch. 22.SE - Prob. 39APCh. 22.SE - Base treatment of the following , -unsaturated...Ch. 22.SE - Prob. 41APCh. 22.SE - Prob. 42APCh. 22.SE - Prob. 43APCh. 22.SE - Which, if any, of the following compounds can be...Ch. 22.SE - Prob. 45APCh. 22.SE - Prob. 46APCh. 22.SE - Prob. 47APCh. 22.SE - How might you convert geraniol into either ethyl...Ch. 22.SE - Prob. 49APCh. 22.SE - One way to determine the number of acidic...Ch. 22.SE - Prob. 51APCh. 22.SE - Prob. 52APCh. 22.SE - Prob. 53APCh. 22.SE - Prob. 54APCh. 22.SE - Prob. 55APCh. 22.SE - Prob. 56APCh. 22.SE - All attempts to isolate primary and secondary...Ch. 22.SE - How would you synthesize the following compounds...Ch. 22.SE - Prob. 59APCh. 22.SE - Prob. 60APCh. 22.SE - Prob. 61APCh. 22.SE - Prob. 62APCh. 22.SE - As far back as the 16th century, South American...Ch. 22.SE - The key step in a reported laboratory synthesis of...Ch. 22.SE - Prob. 65AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Structure pK, K. Name NH; CHCH,-O 2.20 (СО,Н) 9.31 (NH,) 6.3 X 10-3 4.9 x 10-10 Phenylalanine CO,H 7.11 x 10-3 6.34 x 10-8 2.148 * 7.198 Phosphoric acid* (hydrogen phosphate) НО — Р— Он 12.375 4.22 X 10-13 OH (1.5) 6.78 3 x 10-2 1.66 x 10-7 HP- он Phosphorous acid (hydrogen phosphite) OH 1.12 x 10-3 3.90 x 10-6 2.950 Phthalic acid (benzene-1,2-dicarboxylic acid) 5.408 cO,H Piperazine (perhydro- 1,4-diazine) 5.333 9.731 4.65 x 10-6 1.86 X 10-10 11.125 7.50 x 10-12 Piperidine 1.12 x 10-2 2.29 x 10-11 -co,H 1.952 (СO,Н) 10.640 (NH,) Prolinc +H 4.874 1.34 x 10-5 Propanoic acid CH,CH,CO,H H,C=CHCO,H 4.258 5.52 x 10-5 Propenoic acid (аcrylic acid) 10.566 2.72 x 10-11 Propylamine CH;CH,CH,NII; 5.20 6.3,X 10-6 Pyridine (azine) Pyridine-2-carboxylic acid (picolinic acid) (1.01) (CO,H) 5.39 (NH) 9.8 x 10-2 4.1 x 10-6 co,H HO,C Pyridine-3-carboxylic acid (nicotinic acid) 2.03 (CO,H) 4.82 (NH) 9.3 x 10-3 1.51 x 10-s NH+ 1.4 (POH) (u = 0.1) 3.44 (OH) (1p. = 0.1) 6.01 (POH) (u = 0.1) 8.45 (NH) (u =…arrow_forwardmolecular weight of H2O is 18 but his boilling point 100C, WHY? *arrow_forwardA) Phenol Red is a weakly acidic dye which is yellow in the HA form and red in the A form. What concentration of Phenol Red is required to obtain an absorbance of 0.50 at 550 nm at pH 7.8? Note: ε550 of A = 2.5 x 104 M-1 cm-1; ε550 of HA = 0; pKa = 7.8. B) What would the absorbance of the solution in part (a) be if the pH was 8.8?arrow_forward
- Pls help identify these :(arrow_forward(a) (b) (c) C6H5NHCH3 + HC1 (CH3CH₂)2NH + HNO3 IN CH5NH2 NH₂ AICI 3 1) LiAlH₂/H₂O 2) ethyl acetate + H3PO3 2 HCI + NaNO₂ 2 H₂O NaCl HCI OH 1. HNO3, H₂SO4 2. Zn(Hg), HCIarrow_forwardCompound 47 is a high-boiling liquid (boiling point 204-205 'C), that reacts with Ag* ion in aqueous ammonia to form a "silver mirror" (the Tollens test). The Mass, IR, and 'H NMR spectra, along with 13C NMR data, are given below. Elemental Analysis: C, 79.97; H, 6.71; O, 13.32.arrow_forward
- The following molecules and ions are grouped by similar structures. Classify each as aromatic, antiaromatic, or nonaromatic. For the aromatic and antiaromatic species, give the number of pi electrons in the ring. (а) нн H H (b) H H H H H H H H (c) (d) H H +, + H H (e) (f) H, H. H (h) CH, H H H. B.arrow_forward3. The pKa1 and pKa,2 of ortho and para isomers of phthalic acid (diprotic acid) are shown below: pKa1 pKa2 phthalic acid terephthalic acid 2.98 5.28 3.51 4.82 CO2H CO2H HO2C phthalic acid terephthalic acid Provide an explanation on the differences of their pKa values.arrow_forward[1] LIAIH4 [2] H2O [1] [2] H2O `Mg H2 Pd-C [1] LDA, THF,-78°C [2]arrow_forward
- (2p) General Instruction: Give a brief interpretation from the table in the picture. Provide or answer also the ‘Descriptive Equivalent’ of the table. Make sure the interpretation is related to the given question in the table. Please refer to the subsequent picture for your guide. Note: Make sure your interpretation is the same or similar format with the guide provided that is shown in the subsequent picture . Title for the tables: Youth Awareness and Perception Towards COVID-19. Legend: SD – Strongly Disagree D – Disagree A – Agree SA – Strongly Agreearrow_forwardPlease help with 4c, 4d, and 4e. We havent learned kaw of equivalents so please do 4c a different way. thank youarrow_forwardPrioritize these groups as 1,2,3 &4 with 1 being highest priority.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY