
Concept explainers
Interpretation:
Oxidation and reduction should be described and also the electron transfer in a redox reaction and electron donation in a Lewis acid-base reaction has to be compared.
Concept Introduction:
Redox reaction and Half-reaction
- The redox reaction is the combination of
oxidation and reduction reactions and these two reactions are called half-reactions.
Oxidation reaction:
- The loss of electrons or the gain of oxygen atoms is called as oxidation.
Reduction reaction:
- The gaining of electrons or addition of hydrogen atoms is called as reduction.
Explanation of Solution
Oxidation is the reaction in which loss of electron occurs and reduction reaction is the reaction in which gain of electron occurs. Redox reaction is a common term used for a reduction and an oxidation reaction.
In a redox reaction, oxidation always accompanies reduction. Thus one atom must lose electrons and other must gain electrons.
Oxidation reaction:
In the above reaction, Ag atom loses one electron and changes their oxidation state 0 to 1.
Reduction reaction:
In the above reaction
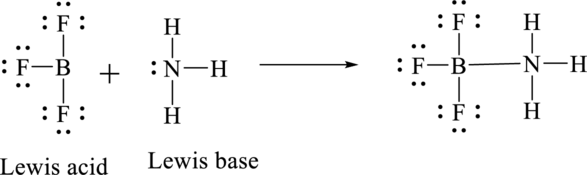
According to Lewis concept of acid and bases,
Lewis acid is an electron pair acceptor.
Lewis base is an electron pair donor.
Want to see more full solutions like this?
Chapter 18 Solutions
Chemistry: Principles and Practice
- A copper penny can be dissolved in nitric acid but not in hydrochloric acid. Using reduction potentials from your textbook, show why this is so. What are the products of the reaction?arrow_forward6. Acids and bases are defined by their donation or acceptance of a proton according to the (a) Arrhenius model. (b) Bronsted-Lowry model. (c) Lewis model.arrow_forwardWhen excess dilute hydrochloric acid is added to a mixture of calcium oxide and magnesium oxide. A highly exothermic neutralisation reaction occurs.Name two salts which are present in the mixture after the reactionarrow_forward
- Describe the process of Electrolysis of water?arrow_forwardOxygen gas has an oxidizing strength comparable to that of nitric acid . Patina is a green or greenish -blue coating that forms on copper metal in the environment . Account for its formationarrow_forwardDefine ionization enthalpy and electron gain enthalpy?arrow_forward
- If the given statement is true, write T. Otherwise, write F and explain why it is false. 1) Equilibrium happens for reactions that are reversible. 2) In the electrolysis of water, the electrical energy from a battery can "pull" electrons from oxygen, and "push" electrons to hydrogen.arrow_forwardSulfurous acid (H,SO,) is a polyprotic acid. Write balanced chemical equations for the sequence of reactions that sulfurous acid can undergo when it's dissolved in water.arrow_forwardIs bromate ion an oxidizing or reducing agent?arrow_forward
- During one of the washes, 10 mL of dilute NaOH was added to the product mixture. Select the statements that are true. NaOH is neither acidic nor basic, but neutral, which is characteristic of sodium compounds. The purpose of the NaOH is to neutralize H,SO, that is present in the mixture in an acid-base reaction. The purpose of the NaOH is to absorb water in the product mixture.arrow_forwardHypobromous Acid, HOBr, and hypoiodous acid, HOI are both weak acids. Which acid is stronger and why? You may look up the correct Lewis structure for these acids to help you answer the question.arrow_forwardCalculate the hydrogen ion concentration in mol/L for solutions with the following pH values: (a) 2.42, (b) 11.21, (c) 6.96, (d) 15.00.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning



