
Concept explainers
(a)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
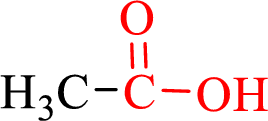
Nomenclature of
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the
alkane name with –oic acid.
Naming of compounds with two
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
(a)
Explanation of Solution
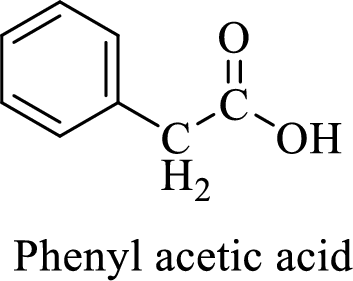
Name of the given compound is phenyl acetic acid.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a carboxylic acid.
- ✓ A phenyl substituent is attached to the acetic acid; Acetic acid is the common name of ethanoic acid.
Thus,
The structural formula for this compound can be drawn as shown below,
(b)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
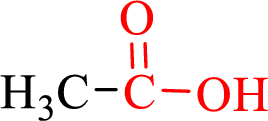
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
(b)
Explanation of Solution
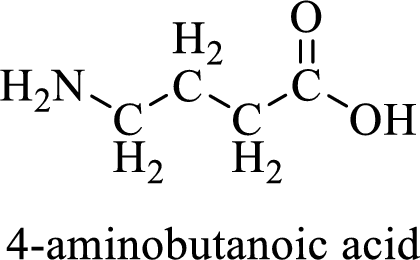
Name of the given compound is 4-Aminobutanoic acid
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a carboxylic acid.
- ✓ Parent chain contains four carbon atoms including the carboxylic acid functional group.
- ✓ An amino substituent is attached to the fourth carbon atom in the parent chain.
Thus,
The structural formula for this compound can be drawn as shown below,
(c)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
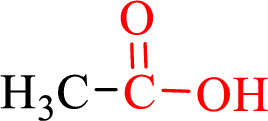
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
(c)
Explanation of Solution
Name of the given compound is 3-Chloro-4-phenylbutanoic acid.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a carboxylic acid.
- ✓ Parent chain contains four carbon atoms including the carboxylic acid functional group.
- ✓ One chlorine atom and a phenyl ring substituents are attached to the third and fourth carbon atoms in the parent chain respectively.
Thus,
The structural formula for this compound can be drawn as shown below,
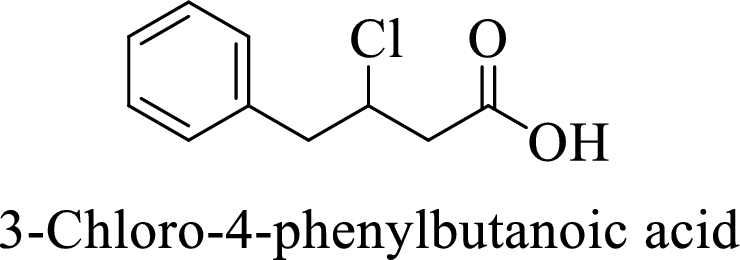
(d)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
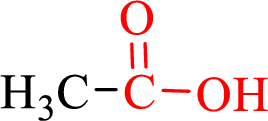
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
(d)
Explanation of Solution
Name of the given compound is Propenoic acid (acrylic acid).
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a carboxylic acid.
- ✓ Parent chain contains three carbon atoms including the carboxylic acid functional group.
- ✓ There is a double bond in the compound.
Thus,
The structural formula for this compound can be drawn as shown below,
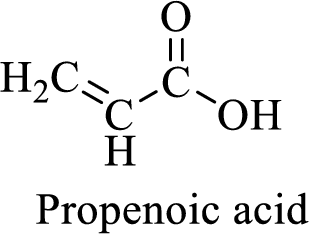
(e)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
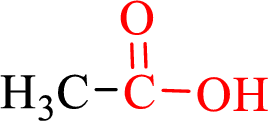
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
E-Z designators are used as like cis-trans terminology for non-similar groups attached
In E-Z designations, the groups attached to vinylic positions are checked by their priority on the basis of higher molecular weight. If the higher priority groups are on the same sides, then the configuration is designated as Z. If the higher priority groups are on the opposite sides, then the configuration is designated as E.
(e)
Explanation of Solution
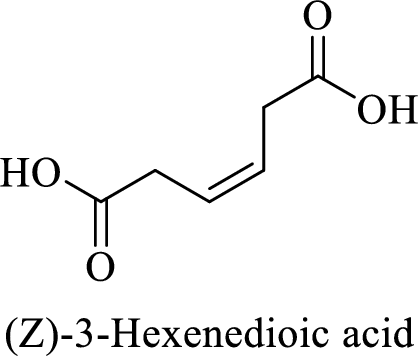
Name of the given compound is (Z)-3-Hexenedioic acid.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a dicarboxylic acid.
- ✓ Parent chain contains six carbon atoms including the carboxylic acid functional group.
- ✓ There is a double bond in the compound. The higher priority substituents are on same side; (Z)
Thus,
The structural formula for this compound can be drawn as shown below,
(f)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
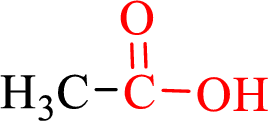
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
(f)
Explanation of Solution
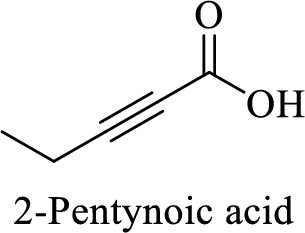
Name of the given compound is 2-Pentynoic acid.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a monocarboxylic acid.
- ✓ Parent chain contains five carbon atoms including the carboxylic acid functional group.
- ✓ There is a triple bond in between second and third carbon atoms in the compound.
Thus,
The structural formula for this compound can be drawn as shown below,
(g)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
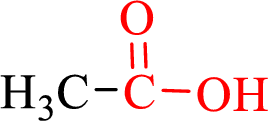
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
(g)
Explanation of Solution
Name of the given compound is Potassium phenylacetate.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a potassium salt of phenyl acetic acid.
- ✓ A phenyl substituent is attached to the acetic acid; Acetic acid is the common name of ethanoic acid.
Thus,
The structural formula for this compound can be drawn as shown below,
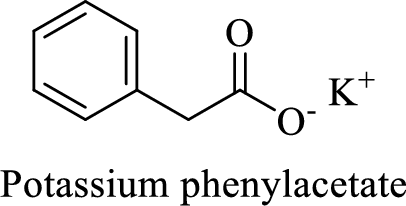
(h)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
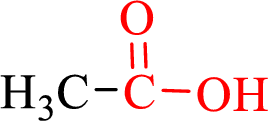
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
(h)
Explanation of Solution
Name of the given compound is sodium oxalate.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a sodium salt of oxalic acid.
- ✓ Two sodium atoms are attached to the both carboxyl group of oxalic acid.
Thus,
The structural formula for this compound can be drawn as shown below,
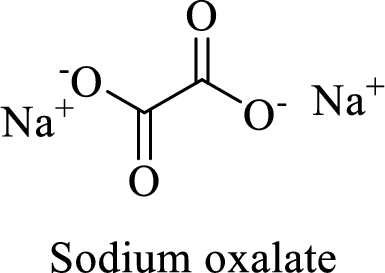
(i)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
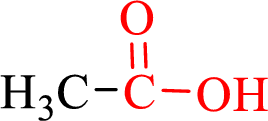
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
(i)
Explanation of Solution
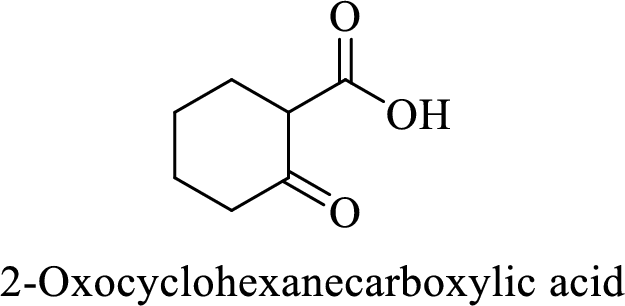
Name of the given compound is 2-Oxocyclohexanecarboxylic acid.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a monocarboxylic acid.
- ✓ Parent ring contains six carbon atoms including the carboxylic acid functional group.
- ✓ There is a
ketone group on second carbon atom in the parent ring.
Thus,
The structural formula for this compound can be drawn as shown below,
(j)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
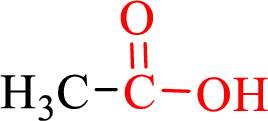
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
(j)
Explanation of Solution
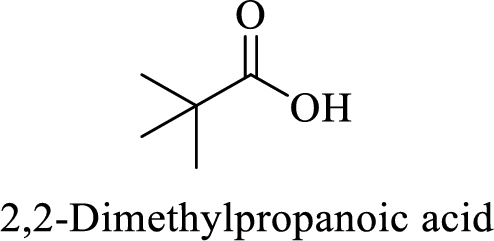
Name of the given compound is 2, 2-Dimethylpropanoic acid.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a monocarboxylic acid.
- ✓ Parent chain contains three carbon atoms including the carboxylic acid functional group.
- ✓ Two methyl substituents are attached on the second carbon atom in the parent chain.
Thus,
The structural formula for this compound can be drawn as shown below,
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry
- A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.(a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyoxygen atom.(b) Compare the resonance forms with those given previously for an acid protonated on thecarbonyl oxygen atomarrow_forwardDraw each molecule given its name and the following information. (a) Nitroglycerin, also known as 1,2,3-trinitroxypropane, the active ingredient in dynamite and a medication administered to people having a heart attack, (Hint: The nitroxy group is the conjugate base of nitric acid.)arrow_forward(a) Explain how NaBH, in CH;OH can reduce hemiacetal A to 1,4-butanediol (HOCH,CH,CH,CH,OH). (b) What product is formed when A is treated with Ph;P=CHCH,CH(CH),? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects. PPha NaOCH,CH3 HO- isotretinoin HO A Br X Yarrow_forward
- Draw a structural formula for each amine. (a) 2-Butanamine (b) 1-Octanamine (c) 2,2-Dimethyl-1-propanamine (d) 1,5-Pentanediamine (e) 2-Bromoaniline (f) Tributylaminearrow_forwardHypoglycin A, an amino acid derivative found in unripened lychee, is anacutely toxic compound that produces seizures, coma, and sometimesdeath in undernourished children when ingested on an empty stomach. (a) Draw the neutral, positively charged, and negatively charged forms of hypoglycin A. (b) Which form predominates at pH = 1, 6, and 11? (c) What is the structure of hypoclycin A at its isoelectric point?arrow_forward(a) Compound Z is a tertiary aromatic amine with the formula, C8H11N. Provide a chemical structure for compound Z. (b)nDraw the structure of the product formed exclusively when nitrous acid reacts with Z.arrow_forward
- 1. Draw structures corresponding to the following IUPAC names: (a) 4-Methylpentanoic acid (b) o-Hydroxybenzoic acid (c) 2,2-Dimethylpropanoyl chloride (d) trans-2-Methylcyclohexanecarboxamide (e) p-Methylbenzoic anhydride (f) p-Bromobenzonitrilearrow_forwardComplete the equation to show how pyridine, C, H, N, acts as a Brønsted-Lowry base in water. equation: C₂H₂N+H₂O=arrow_forwardb) Compounds in which the –OH of the carboxyl group is replaced by certain other groups are called carboxylic acid derivatives. The most important of which are acyl halides, acid anhydrides, esters and amides. Discuss the relationship between carboxylic acid, acid chloride and acid anhydridearrow_forward
- Given that C6H11COOH has a pKa = 4.8 and C6H11N+H3 has a pKa = 10.7, (a) What pH would you make the water layer to cause the carboxylic acid to dissolve in the water layer and the amine to dissolve in the ether layer? (b) What pH would you make the water layer to cause the carboxylic acid to dissolve in the ether layer and the amine to dissolve in the water layer?arrow_forward(a) Rank the following compounds in order of increasing acidity. (b) Which compound forms the strongest conjugate base?arrow_forwardDescribe concisely a chemical test to distinguish between the following pairs of compounds.(a) Propanal and propanone(b) Phenol and benzoic acid(c) Hexan-3-one and hexan-2-onearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

