
Concept explainers
(a)
Interpretation:
The chiral center in the following compound should be located.
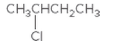
Concept Introduction:
Compounds in which an atom is attached to four different groups of atoms are known as chiral center and the compound is known as a chiral compound.
(b)
Interpretation:
The chiral center in the following compound should be located.
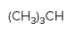
Concept Introduction:
Compounds in which an atom is attached to four different groups of atoms are known as chiral center and the compound is known as a chiral compound.
(c)
Interpretation:
The chiral center in the following compound should be located.
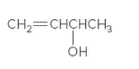
Concept Introduction:
Compounds in which an atom is attached to four different groups of atoms are known as chiral center and compound is known as a chiral compound.
(d)
Interpretation:
The chiral center in the following compound should be located.
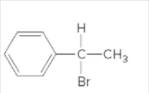
Concept Introduction:
Compounds in which an atom is attached to four different groups of atoms are known as chiral center and compound is known as a chiral compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 3.A carbon atom that is bonded to four different groups is a/an A.achiral center B.chirality center C.symmetrical center D.symmetrical centerarrow_forwardLabel each compound as chiral or achiral. Compounds that contain a single carbon common to two rings are called spiro compounds. Because carbon is tetrahedral, the two rings are perpendicular to each other.arrow_forwardball & stick + labels stereoisomer(s) e. Are the two molecules enantiomers? yes no ball & stick a. How many chiral carbons are there in the molecule on the left? chiral carbon(s) b. What is the total number of stereoisomers in the group that includes the molecule on the left? stereoisomer(s) c. How many chiral carbons are there in the molecule on the right? chiral carbon(s) d. What is the total number of stereoisomers in the group that includes the molecule on the right? + labels Previous Next Save and Exitarrow_forward
- Ephedrine, a stimulant and illegal drug is an optically active compound with two chiral centers. OH NHCH3 CH3 Ephedrine a. In the structure of ephedrine on the answer sheet, mark each chiral center with an asterisk. b. How many stereoisomer/s does ephedrine have? Show necessary solutions on the answer sheet. c. Compound EPS is a stereoisomer of ephedrine. Determine the absolute configuration (R or S) at carbons A and B of compound EPS.arrow_forward5. Cholesterol is one of the important component of animal cell membrane. Label circled chiral centers as R/S, and circle the rest of chiral centers in this compound. b a H НО Cholesterolarrow_forwardLabel the compounds as chiral or achiral. CH3 H3C. H CH3 CHO H- -OH H- OH H- -OH CH₂OHarrow_forward
- A) ОН B) HO. Molecule A is achiral and molecule B is chiral Molecule A is chiral and molecule B is achiral Both molecules are achiral. Both molecules are chiral.arrow_forwardPlace an asterisk (*) next to the chirality center (sometimes called chiral atom) or centers in each compound. It is possible that a compound may not have a chiral center.arrow_forwardConsider CH3-CH(OH)-CH(OH)(Br). a.How many stereogeniccenters are in the molecule? b.How many stereoisomers are there for the compound? c.Draw the Fischer projection for each of the stereoisomer. Label each using I, II, etc. d.Which pairs are enantiomers? Which are diastereomers? e.Determine the absolute configuration of each chiral center in one pair of diastereomer.arrow_forward
- 1a. How many stereogenic centers are present 1c. Draw a three-dimensional structure of a in the structure below? Indicate them with asterisk(s). How many stereoisomers stereoisomers are possible? chiral compound with the molecular formula of C4H4Cl₂ that does not have a stereogenic carbon. In addition, draw the enantiomer of this compound. Number of stereogenic centers: Number of stereoisomers possible: 1b. Draw one of the two most stable stereoisomers of the compound in 1a using a planar structure with wedges and dashes. Now draw it in its preferred chair conformation. 1d. Draw two meso compounds with the molecular formula of C7H14.arrow_forwardModify the existing drawings to draw both stereoisomers of this compound. H₂C₂ CH₂ HO OH H H₂C Edit Drawing CH3 HO OH Harrow_forwardConsider the following molecules (a-h) and answer the questions below:i. How many chiral carbon does it contains?ii. Does the molecule have any internal plane of symmetry?iii. Identify the molecule whether it is chiral of achiral?iv. Does it rotate the plane of polarized light?v. Is the molecule optically active?vi. Is meso compound present?vii. Draw all possible stereoisomers for the molecule in Fischer projection and label the relationship between each of the isomer.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

