
Concept explainers
14-8 Answer true or false.
- The
functional group of an alcohol is the —OH (hydroxyl) group.
(h, The solubility of alcohols in water increases with increasing molecular weight.
(a)
Interpretation:
To analyse whether the given statement: The functional group of an alcohol is the −OH (hydroxyl) group, is true or not.
Concept Introduction:
Alcohols are those organic compounds wherein a hydrogen atom of the aliphatic carbon molecule is substituted by a hydroxyl group. Hence an alcohol molecule has two parts, one comprising the alkyl group and the other carrying hydroxyl group.
The boiling point of alcohols is significantly higher than those of alkanes having similar molecular weights. The solubility of alcohol is inversely proportional to the size of the alkyl group. Alcohols reacts with active metals like sodium, potassium and many more to form the corresponding alkoxide.
Answer to Problem 14.8P
The functional group of an alcohol is the −OH (hydroxyl) group. Thus, the statement is true.
Explanation of Solution
The functional group of any alcohol is the —OH group. Therefore, the given statement is true.
(b)
Interpretation:
To analyse whether the given statement: The parent name of an alcohol is the name of the longest carbon chain that contains the −OH group, is true or not.
Concept Introduction:
Alcohols are those organic compounds wherein a hydrogen atom of the aliphatic carbon molecule is substituted by a hydroxyl group. Hence an alcohol molecule has two parts, one comprising the alkyl group and the other carrying hydroxyl group.
The boiling point of alcohols is significantly higher than those of alkanes having similar molecular weights. The solubility of alcohol is inversely proportional to the size of the alkyl group. Alcohols reacts with active metals like sodium, potassium and many more to form the corresponding alkoxide.
Answer to Problem 14.8P
The parent name of an alcohol is the name of the longest carbon chain that contains the −OH group.Thus, the statement is true.
Explanation of Solution
According to IUPAC nomenclature, the parent name of an alcohol is the name of the longest.
carbon chain that contains the —OH group. Therefore, the given statement is true.
(c)
Interpretation:
To analyse whether the given statement: A primary alcohol contains one −OH group, and a tertiary alcohol contains three-OH groups, is true or not.
Concept Introduction:
Alcohols are those organic compounds wherein a hydrogen atom of the aliphatic carbon molecule is substituted by a hydroxyl group. Hence an alcohol molecule has two parts, one comprising the alkyl group and the other carrying hydroxyl group.
The boiling point of alcohols is significantly higher than those of alkanes having similar molecular weights. The solubility of alcohol is inversely proportional to the size of the alkyl group. Alcohols reacts with active metals like sodium, potassium and many more to form the corresponding alkoxide.
Answer to Problem 14.8P
A primary, secondary, and tertiary alcohol contains only one -OH groups but the connectivity of carbon atoms bearing −OH group is different. Thus, the statement is false.
Explanation of Solution
Primary alcohol is alcohol in which the -OH group is linked to carbon that is connected to only one carbon atom. It has like -CH2 OH group in molecule. Secondary alcohol is alcohol in which the -OH group is linked to carbon that is connected to two other carbon atoms. Tertiary alcohol is alcohol in which the -OH group is linked to carbon that is connected to three other carbon atoms. Primary, secondary, and tertiary alcohols have only one -OH group in their molecule but the connectivity of carbon atom bearing -OH group is different. Therefore, the given statement is false.
(d)
Interpretation:
To analyse whether the given statement: In the IUPAC system, the presence of three −OH groups is shown by the ending −triol, is true or not.
Concept Introduction:
Alcohols are those organic compounds wherein a hydrogen atom of the aliphatic carbon molecule is substituted by a hydroxyl group. Hence an alcohol molecule has two parts, one comprising the alkyl group and the other carrying hydroxyl group.
The boiling point of alcohols is significantly higher than those of alkanes having similar molecular weights. The solubility of alcohol is inversely proportional to the size of the alkyl group. Alcohols reacts with active metals like sodium, potassium and many more to form the corresponding alkoxide.
Answer to Problem 14.8P
In the IUPAC system, the presence of three −OH groups is shown by the ending −triol. Thus, the statement is true.
Explanation of Solution
In IUPAC nomenclature, the presence of three -OH groups is shown by the ending triol Therefore, the given statement is true.
(e)
Interpretation:
To analyze whether the given statement: A glycol is a compound that contains two-OH groups. The simplest glycol is ethylene glycol,
Concept Introduction:
Alcohols are those organic compounds wherein a hydrogen atom of the aliphatic carbon molecule is substituted by a hydroxyl group. Hence an alcohol molecule has two parts, one comprising the alkyl group and the other carrying hydroxyl group.
The boiling point of alcohols is significantly higher than those of alkanes having similar molecular weights. The solubility of alcohol is inversely proportional to the size of the alkyl group. Alcohols reacts with active metals like sodium, potassium and many more to form the corresponding alkoxide.
Answer to Problem 14.8P
A glycol is a compound that contains two-OH groups. The simplest glycol is ethylene glycol,
Explanation of Solution
Glycol is a compound that has two -OH groups attached to carbon atoms. Ethyl glycol is the simplest form of glycol. Its structural formula is:
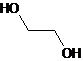
Therefore, the given statement is true.
(f)
Interpretation:
To analyze whether the given statement: Because of the presence of an-OH group, all alcohols are polar compounds, is true or not.
Concept Introduction:
Alcohols are those organic compounds wherein a hydrogen atom of the aliphatic carbon molecule is substituted by a hydroxyl group. Hence an alcohol molecule has two parts, one comprising the alkyl group and the other carrying hydroxyl group.
The boiling point of alcohols is significantly higher than those of alkanes having similar molecular weights. The solubility of alcohol is inversely proportional to the size of the alkyl group. Alcohols reacts with active metals like sodium, potassium and many more to form the corresponding alkoxide.
Answer to Problem 14.8P
Because of the presence of an -OH group, all alcohols are polar compounds. Thus, the statement is true.
Explanation of Solution
Polar molecules have polar covalent bonds connected between the atoms. Methanol is an example of alcohol. Here, the O-C bond and O-H bond are polar covalent bonds. In methanol, there is a difference in electronegativity between the O-C bond (3.5-2.5 = 1.0) and O-H bond (3.5-2.1 = 1.4) Electronegativity is the tendency of an atom to attract electrons towards itself. Due to the presence of the O-H bond, all alcohols are polar molecules.
Therefore, the given statement is true.
(g)
Interpretation:
To analyze whether the given statement: The boiling point of alcohols increases with increasing molecular weight, is true or not.
Concept Introduction:
Alcohols are those organic compounds wherein a hydrogen atom of the aliphatic carbon molecule is substituted by a hydroxyl group. Hence an alcohol molecule has two parts, one comprising the alkyl group and the other carrying hydroxyl group.
The boiling point of alcohols is significantly higher than those of alkanes having similar molecular weights. The solubility of alcohol is inversely proportional to the size of the alkyl group. Alcohols reacts with active metals like sodium, potassium and many more to form the corresponding alkoxide.
Answer to Problem 14.8P
The boiling point of alcohols increases with increasing molecular weight. Thus, the statement is true.
Explanation of Solution
The temperature at which the atmospheric pressure is equal to the vapor pressure is known as boiling point. As the molecular weight increases, more atoms are involved in the non-covalent interaction. Due to this tact, more energy (more temperature) is required to break this non-covalent interactions.Therefore, the given statement is true.
(h)
Interpretation:
To analyze whether the given statement: The solubility of alcohols in water increases with increasing molecular weight, is true or not.
Concept Introduction:
Alcohols are those organic compounds wherein a hydrogen atom of the aliphatic carbon molecule is substituted by a hydroxyl group. Hence an alcohol molecule has two parts, one comprising the alkyl group and the other carrying hydroxyl group.
The boiling point of alcohols is significantly higher than those of alkanes having similar molecular weights. The solubility of alcohol is inversely proportional to the size of the alkyl group. Alcohols reacts with active metals like sodium, potassium and many more to form the corresponding alkoxide.
Answer to Problem 14.8P
The solubility of alcohols in water decreases with increasing molecular weight. Thus, the statement is false.
Explanation of Solution
Alcohols are more soluble in water than hydrocarbons. This is due to the presence of the O-H group in alcohol. The O-H group undergoes hydrogen bonding with a hydrogen atom in the water molecule.
As the molecular weight increases, simultaneously the hydrocarbon chain in alcohol will also increase. Now the alcohol stars behaving more like hydrocarbons. Therefore, the solubility decreases with the increase in molecular weight.
Therefore, the given statement is false.
Want to see more full solutions like this?
Chapter 14 Solutions
Introduction to General, Organic and Biochemistry
- Microsoft Edge X + een%20kaur/Downloads/1015MSC%20T1%202022%20Lab%20Workbook.pdf CD Page view A Read aloud TAdd text Draw Highlight 4. Tylenol also is an analgesic often taken by people allergic to aspirin. The active ingredient is acetaminophen. HO -NH FO H3C Would acetaminophen give a positive phenol test? (Y/N). Does this compound contain the ester functional group? If not, what functional group(s) is/are present? 10 Sarrow_forwardMixing cyclohexanol with phosphoric acid is an exothermic process, whereas the production of cyclohexene is endothermic. Construct an energy diagram showing the course of this reaction. Label the diagram with the starting alcohol, the oxonium ion (the protonated alcohol), the carbocation, and the product.arrow_forwardQuestion 18.c of 20 Aldehydes and ketones are common carbonyl functional groups. Identify the structures and provide names of these molecules. Provide a systematic name for this structure. سکتے 5- H 1- 4- cyclo tert- 6- di n- tri 3- 2- sec- hex eth but Submit +arrow_forward
- 18. In the IUPAC name for the following compound, the -Br group is located on carbon number CH;CHCH=CH, Br a. 1 b. 2 С. 3 d. 4arrow_forwardOxidation and Reduction of Aldehydes and Ketones (Section 15-10) 15-75 Draw the structural formula of the carboxylic acid formed when each of the following aldehydes is oxidized. a. CH,-C-H CH3 b. CH;-CH,-ČH-CH,-C-H с. Н-С—Н ÇI ÇI d. CH;—CH,—CH-CH—CH2—С-нarrow_forwardA solid sticky substance which strongly repels water is made of the following molecules: CH3 (CH2)5 — || (CH2)14-CH3 If it is treated with sulfuric acid and heat, two new substances can be recovered. One is an alcohol. Write the chemical formula of the alcohol. Give the common (not IUPAC) name of the other substance. 0arrow_forward
- Description Solubility in water (+/-) Sample Skeletal Structure (i.e., physical state, odor, clarity and color) Ethanol 1-Butanol Cyclohexanol Isoamyl alcohol Phenoliolpgy Environmental Science Explain in 2 – 3 sentences the solubility of alcohols and phenols in water.arrow_forward12-50 Draw the structural formula of an alkene that undergoes acid-catalyzed hydration to give the indicated alcohol as the major product. More than one alkene may give each alcohol as the major product. 3-Hexanol 1-Methylcyclobutanol 2-Methyl-2-butanol 2-Propanolarrow_forward16-17 Propylamine (bp 48°C), ethylmethylamine (bp 37°C), and trimethylamine (bp 3°C) are constitutional isomers with the molecular formula C3HgN. Account for the fact that trimethylamine has the lowest boiling point of the three and propylamine has the highest.arrow_forward
- A E F * Change the units within the perentheses to the ones you are using! We will need these to label your graph axes MWt (units Boiling point (units Compound name #C atoms go here) go here) 3 ethane 4 propane 5 butane 6 pentane 7 hexane 8 ethanol 9 1-propanol 10 1-butanol 11 1-pentanol 12 1-hexanol 13 ethanal 14 propanal 15 butanal 16 pentanal 17 hexanal 18 ethanamine 19 propanamine 20 butanamine 21 pentanamine 22 hexanamine 23 ethanoic acid 24 propanoic acid 25 butanoic acid 26 pentanoic acid 27 hexanoic acidarrow_forwardRank the following compounds by their boiling point. Use 1-6 with 6 being the highest. A B C E F ОН E ОН LL B ОНarrow_forward= A ALEKS-Jacari Johnson O ORGANIC FUNCTIONAL GROUPS Naming and drawing alcohols Write the systematic (IUPAC) name for each of the following organic molecules: structure OH HO–CH2C—CH…CH3 CH3OH- OH OH CH3-CH-CH₂-C-CH₂-OH CH3 OH name 0 0 0arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

