
Concept explainers
(a)
Interpretation:
Whether the
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. The phenomenon is called isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 12.18E
The alkene
Explanation of Solution
The alkene is
A compound shows cis-trans isomerism when the substituent groups on the carbon atoms that are connected via double bond are different. The alkene has the same substituted groups on a carbon atom that is on the right-hand side of the double bond. Therefore; it will not have cis-trans isomerism.
The alkene
(b)
Interpretation:
Whether the alkene
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. This phenomenon is called isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 12.18E
The alkene
Explanation of Solution
The alkene is
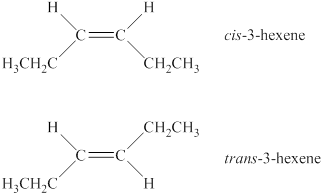
A compound shows cis-trans isomerism when the substituents groups on the carbon atoms that are connected via double bond are different. The alkene has two different substituted groups on carbon atoms that are connected via a double bond. Therefore, it will show cis-trans isomerism. In the cis isomer, same substituted groups are on the same side of a ring or double bonds, whereas in the trans isomer same substituted groups are on a different side of a ring or double bond. Therefore, the structures of cis-trans isomers of the alkene are shown below.

Figure 1
The alkene
(c)
Interpretation:
Whether the alkene
Concept introduction:
Those compounds which have the same molecular formula but have different arrangements of atoms are known as isomers. The phenomenon is called isomerism. The isomers are generally classified as structural isomers and stereoisomers. Stereoisomers are further divided into two categories diastereomers and enantiomers.
Answer to Problem 12.18E
The alkene
Explanation of Solution
The structure of the alkene
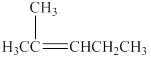
Figure 2
A compound shows cis-trans isomerism when the substituents groups on the carbon atoms that are connected via double bond are different. The alkene has the same substituted groups on a carbon atom that is on the left-hand side of the double bond. Therefore, it will not have cis-trans isomerism.
The alkene
Want to see more full solutions like this?
Chapter 12 Solutions
Chemistry for Today: General, Organic, and Biochemistry
- What is a hydrocarbon? What is the difference between a saturated hydrocarbon and an unsaturated hydrocarbon? Distinguish between normal and branched hydrocarbons. What is an alkane? What is a cyclic alkane? What are the two general formulas for alkanes? What is the hybridization of carbon atoms in alkanes? What are the bond angles in alkanes? Why are cyclopropane and cyclobutane so reactive? The normal (unbranched) hydrocarbons are often referred to as straight-chain hydrocarbons. What does this name refer to? Does it mean that the carbon atoms in a straight-chain hydrocarbon really have a linear arrangement? Explain. In the shorthand notation for cyclic alkanes, the hydrogens are usually omitted. How do you determine the number of hydrogens bonded to each carbon in a ring structure?arrow_forwardSelect those compounds that can be correctly called unsaturated and classify each one as an alkene or an alkyne: a.CH3CH2CH3f. b.CH3CH=CHCH3g. c.h.CH2=CHCH2CH3 d.i. e.arrow_forwardGasohol is a mixture of 90% gasoline and 10% ethanol, CH 3CH 2OH. Ethanol is considered an environmentally friendly fuel additive because it can be made from a renewable source—sugarcane. Ethanol burns in air to form CO 2 and H 2O, and, like the combustion of alkanes, this reaction also releases a great deal of energy. Write a balanced equation for the combustion of ethanol.arrow_forward
- Decane has the structure: CH3(CH2)9CH3. What does the prefix "dec-" specifically indicate about the structure of this compound? Multiple Choice It contains a straight chain of carbons. It contains 10 carbons in a continuous chain. It is an alkane. It is a saturated hydrocarbon.arrow_forward1. Using the grignard reaction of alkanes what is the resulting alkane if the reactant is C4H9Br? a. ethane b. propane c. butane d. pentane 2. Using the grignard reaction of alkanes what is the resulting alkane if the reactant is C5H11F? a. ethane b. propane c. butane d. pentane 3. Using Cl2 in C2H4Cl2 will result in HCl and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. not posiblearrow_forwardGeneral formula of alkenes is a. CnH2n-2 b. CnH2n c. Cn-2Hn d. CnH2n+2arrow_forward
- Classify the following hydrocarbons as alkanes, cycloalkane, alkene, cycloalkenes, alkyne, cycloalkyne, or aromatic.a. CH2 CH CH2b. CH3 C (CH3)2 CH (CH2CH3) CH2 CH2 CH (CH3)2c. (CH3)3 C C C CH (CH3) CH2 CH3arrow_forward13. Ethylethanoate and butanoic acid can be classified as A. positional isomers B. chain isomers C. functional isomers D. stereoisomers 14. Which of the following pairs are positional isomers A. trans-1,4-dichlorocyclohexane, cis-1,3-dichlorocyclopentane B. trans 1,4-dichlorocyclohexane, cis-1,4-dichlorocyclohexane C. 2-pentanol, Cyclopentanol D. 1,2-cycohexanediol, 1,3-cycohexanediol 15. Which of the following compounds will have zero dipole moment? A. cis-1,2-dibromoethylene B. 1,1-dibromoethylene C. trans-1,2-dibromoethylene D. all of these 16. Which of the following is not aromatic: A. cyclopentadienyl cation B. cyclopentadienyl anion C. Cyclopropenyl cation D. Cycloheptatrienyl cation 17. Which of the following compounds containing lone pair has the least tendency to donate its electrons? A. the lone pair in pyridine B. the lone pair in furan C. the lone pair in pyrole D. the lone pair in thiophenearrow_forwardConsider alkene 3. alkene 3 name: CH2 H2C C. In CH2 H3Carrow_forward
- Based on the molecular formula, determine whether each compound is an alkane, alkene, or alkyne. (Assume that the hydro- carbons are noncyclical and there is no more than one multiple bond.) a. C5H12 b. C3H6 с. С-Н12 d. C11H22arrow_forward1. Which of the following formulas is incorrect?a. CaNO3b. MgSc. AlBr3d. Li2Oe. NaOH 2. Part of an organic molecule where most of its chemical reactions occur.a. Substrateb. Productc. Reactantsd. Functional Group 3. An acyclic unsaturated hydrocarbon that contains one or more carbon-carbon triple bonds. As the family name alkyne indicates the characteristics ending associated with a triple bond. It has a general formula of one triple bond is CnH2n-2.a. Alkenesb. Alkynesc. Aromatic compoundd. Cycloalkanearrow_forward1.Draw the structural formula of CH 3 CH(CH 3 )(CH 2 ) 4 CH3 2. How does the general formula of a cycloalkane compare to that of an alkane?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





