
Pushing Electrons
4th Edition
ISBN: 9781133951889
Author: Weeks, Daniel P.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 20EQ
Using the method outlined above, derive the structures for the following compounds. The unbounded skeletons are provided.
Propyne
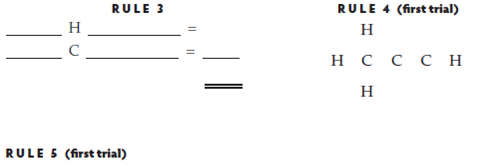
No. of electrons in structure _____
No. of valence electrons _____
Structure is ___________ (correct, incorrect)
(Second trial) (third trial)
______ ______
___________________ ______ _____________________ ______
Structure is _____________ Structure is __________________
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Draw structural formulas for organic products A and B in the window below.
-CI
Li
pentane
H₂O
A
B
•
Draw only products having the organic portion of the original alkyl halide.
•
Draw carbon-lithium bonds using the single bond tool. If a structure has a copper-lithium bond, do not draw the lithium.
•
Separate products from different steps using the →>> sign from the drop-down menu.
?
n [
ChemDoodleⓇ
What are the three groups of organic compounds that contain only carbon and hydrogen? Explain the
difference between the compounds in these three groups.
Use the paperclip button below to attach files.
I U
Ω
acer
@
%24
%
&
4.
08.
9.
e
gh
b n m
alt
Please state the observation for
Chemical properties of Hydrocarbons:
Reaction with KMnO4:
1. Label three clean, dry test tubes with the name of the substance to be tested.
2. Place 4 drops of the sample to be tested in a test tube, carefully add 1 drop of 1% alkaline KMnO4.
3. Record your observations.
4. Disposal: All contents to be disposed into the waste container marked as organic waste.
Wash the test tubes and clean them using brush, soap and water. Keep these inverted in the test tube rack.
Data Table:
Test tube
Observations
1) Alkane
2) Alkene
3) Aromatics
Chapter 1 Solutions
Pushing Electrons
Ch. 1 - 1. Hydrogen is a Group I element and each...Ch. 1 - Methanol has the molecular formula CH4O. Its...Ch. 1 - 3. The skeleton of chloromethane is...Ch. 1 - 4. Methanol’s skeleton is
Connecting all bonded...Ch. 1 - 5. The structure for chloromethane is
It...Ch. 1 - Prob. 6EQCh. 1 - 7. Dimethyl ether
No. of electrons in...Ch. 1 - Methylamine (CH5N) No. of electrons in structure...Ch. 1 - Methanethiol (CH4S) No. of electrons in structure...Ch. 1 - Methylal (C3H8O2) No. of electrons in structure...
Ch. 1 - Prob. 11EQCh. 1 - Adding electrons to the skeleton by making single...Ch. 1 - This is done by removing an unshared pair from...Ch. 1 - Prob. 14EQCh. 1 - Prob. 15EQCh. 1 - Prob. 16EQCh. 1 - The skeleton of acetyl chloride is . Write the...Ch. 1 - Three constitutional isomers exist for the formula...Ch. 1 - A number of constitutional isomers exist for the...Ch. 1 - Using the method outlined above, derive the...Ch. 1 - Prob. 21EQCh. 1 - Prob. 22EQCh. 1 - Prob. 23EQCh. 1 - Prob. 24EQCh. 1 - The skeleton of benzyldimethylamine is
The...Ch. 1 - The skeleton is benzaldoxime is The number of...Ch. 1 - Prob. 27EQCh. 1 - Derive Lewis structures for the compounds below....Ch. 1 - Prob. 29EQCh. 1 - Derive Lewis structures for the compounds below....Ch. 1 - Prob. 31EQCh. 1 - Derive Lewis structures for the compounds below....Ch. 1 - The Lewis structure of acetone is Circling the...Ch. 1 - Chloromethane has the Lewis...Ch. 1 - In the Lewis structure for chloromethane, the...Ch. 1 - Prob. 36EQCh. 1 - The oxygen atom in acetone possesses ____ unshared...Ch. 1 - Nitrobenzene has the skeleton
The number of...Ch. 1 - Prob. 39EQCh. 1 - Compute and add on the formal charges I these...Ch. 1 - Prob. 41EQCh. 1 - Prob. 42EQCh. 1 - Prob. 43EQCh. 1 - Prob. 44EQCh. 1 - Prob. 45EQCh. 1 - Prob. 46EQCh. 1 - Prob. 47EQCh. 1 - Compute and add on the formal charges in these...Ch. 1 - Prob. 49EQCh. 1 - Prob. 50EQCh. 1 - The n-propyl cation can be formed from a molecule...Ch. 1 - Prob. 52EQCh. 1 - Prob. 53EQCh. 1 - Methanol, CH3OH, is a compound in which the formal...Ch. 1 - When a proton becomes bonded to diethyl ether, by...Ch. 1 - Tetrahydrofuran has the structure
When a proton...Ch. 1 - Prob. 57EQCh. 1 - Prob. 58EQCh. 1 - The structure of pyridine is
When a proton...Ch. 1 - The carbon atom owns one electron from each of ...Ch. 1 - The n-butyl anion can be formed from When the CLi...Ch. 1 - The isobutyl anion can be formed from When the CNa...Ch. 1 - Prob. 63EQCh. 1 - Ethanol, , is a compound in which the formal...Ch. 1 - The loss of a proton attached to the oxygen atom...Ch. 1 - A very strong base can remove a proton from...Ch. 1 - Prob. 67EQCh. 1 - Prob. 68EQCh. 1 - Prob. 69EQCh. 1 - The homolysis of the OO bond in diacetyl peroxide...Ch. 1 - Prob. 71EQCh. 1 - Prob. 72EQCh. 1 - Prob. 73EQCh. 1 - Prob. 74EQCh. 1 - Prob. 75EQCh. 1 - Heterolytic cleavage of the CO bond to yield a...Ch. 1 - Prob. 77EQCh. 1 - Prob. 78EQCh. 1 - Prob. 79EQCh. 1 - Prob. 80EQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- b Chemistry x Co. How to Ca x NNSU Logir x PeriodicTa X M Action Rec x 0 mySigTau x G 4.25 ml to x NBA Final X G scientific c X + ow.com/ilrn/takeAssignment/takeCovalentActivity.do?locator=assignment-take [References] Butene, used as the monomer for polybutene, is composed of 85.7% C and 14.3% H by mass. If the density of butene is 2.50 g/L at STP, what is the molecular formula of butene? Submit Answer Try Another Version 2 item attempts remaining tedarrow_forward= Organic Molecules Assignment Draw the structure of iodobenzene. Check Click and drag to start drawing a structure. Terms of Use | Privacy Center Accessibility C™ X A ċ Save For Later tu Submit Assignmentarrow_forwardUsing two monomers only (reactants). Each one of these two monomers should have two functional groups of Carboxyl on the right side and Hydroxyl on the left side attached to them. Hint: The total number of carbon atoms for each one of these two monomers, including the one for the carboxyl, must be four (4) only. a) Draw the proper polymer structure of the products. b) Name the products.arrow_forward
- The following chemical equation is for an addition reaction, where the important atoms, groups, and bonds involved have been highlighted for you: 4 + +CI-CI Use the information provided to predict the missing organic product and draw its structure in the drawing area below. Make sure to use the same skeletal ("line") style for the structure as the rest of the equation. Explanation ? Click and drag to start drawing a structure. Check Ⓒ2023 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibility 577 Se MAYarrow_forwardOrg. Chemistry - CANNOT be hand-drawn! Functional Groups illustrate the structural formula of your molecule (expanded or condensed) and indicate the functional groups present by highlighting, circling, or color-coding each and labeling the group. See my attached image - I need help correcting this with a new computerized illustration Thank you!arrow_forwardPlease explain why only structure #2 is correct. Thank you so much!arrow_forward
- Question 23 Identify the organic class to which the compound belongs based on the functional groups present. [Hint - Organic class such as Ester, Ether, Carboxylic acid, Aldehyde, Ketone, Amine, Amide, Alcohols, if more than on Functional group present identify both, left then right separated by;] H3C-O Paragraph Previous Page CH3 B I U A Next Page |||||| 58 8⁰ + v 11. Page 23 of 30arrow_forwardComplete the following equations. ? ОНarrow_forwarda. Use bond energies (see Table 3-3) to estimate H for the reaction of two molecules of glycine to form a peptide linkage. b. Would you predict S to favor the formation of peptide linkages between two molecules of glycine? c. Would you predict the formation of proteins to be a spontaneous process?arrow_forward
- ssignment/takeCovalentActivity.do?locator=Dassignment-take Delete paystubs-... PaperHelp Great Lakes Login... GAGE School/Work Email [Review Topics] Use the References to access important values if Name each of the following compounds: structural formula name CH2 CH CH2 CH2- CH2 CH3-CH2-CH2-CH2-CHCH3 CH2. CCH2 CH Submit Answer Retry Entire Group 9 more group attempts remainingarrow_forwardtwo of these statements are truths and one is a lie. Which of the following statements is a lie? Select one: a. The human blood follows the aspect of suspension because it contains different sizes of molecules. b. The proton concentration of seawater is 0.00000001 M therefore it is an acidic solution. c. Not all inorganic compounds do not have carbon atoms. Thus it is easier to say that these types of compounds, if compared with organic compounds, are not solely based on composition. guess of the student is Aarrow_forwardThe reason for the development of polycrystalline material is because ( one sentence only)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License