
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.6, Problem 1Q
Acid-Base Equilibria
Many factors contribute to the acidity of organic compounds. Electronegativity, resonance, induction, hybridization,
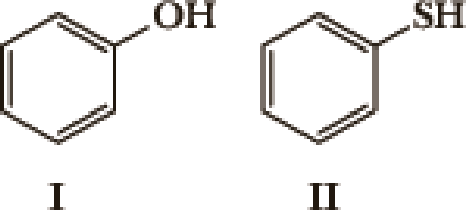
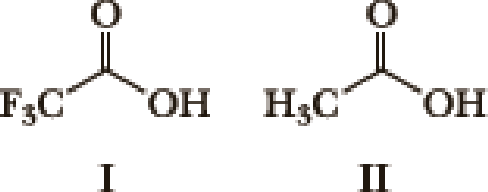
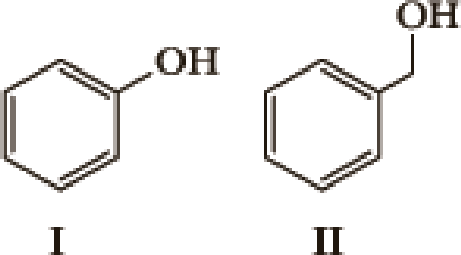
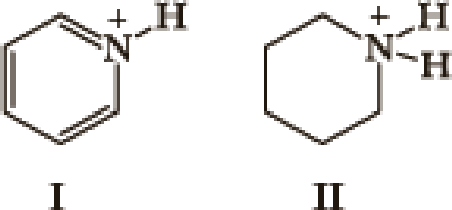
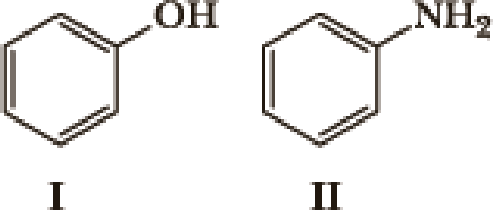
- 1. What factor(s) are the most important to consider when predicting the relative acidity of the two compounds?
- a. Electronegativity of the atom possessing the hydrogen.
- b. Resonance stabilization of the anionic conjugate base.
- c. Inductive stabilization of the anionic conjugate base.
- d. Hybridization of the atom possessing the hydrogen.
- e. The atomic size of the atom possessing the hydrogen.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Provide the bond line structures of two acids, one whose conjugate base is resonance stabilized and one which is not. Explain the effect that resonance will have on acidity.
Arrange the given molecules by increasing acidity. Elaborate these in terms of their partial charges and their inductive effects.
The following table shows the structures of some organic molecules together with their pKa values.Write the ionization reaction, they must take into account the hydrogen that is given off, the hydrogen acid. What is the strongest acid? Propose an explanation to justify the difference in acidity values.
Chapter 4 Solutions
Organic Chemistry
Ch. 4.2 - For each conjugate acid-base pair, identify the...Ch. 4.2 - Write these reactions as proton-transfer...Ch. 4.2 - Following is a structural formula for guanidine,...Ch. 4.2 - Write an equation to show the proton transfer...Ch. 4.3 - For each value of Ka, calculate the corresponding...Ch. 4.4 - Predict the position of equilibrium and calculate...Ch. 4.5 - Calculate Keq for a reaction with G0 = 17.1 kJ/mol...Ch. 4.6 - Acid-Base Equilibria Many factors contribute to...Ch. 4.6 - What is the relative trend in acidity and pKa of...Ch. 4.7 - Write an equation for the reaction between each...
Ch. 4 - For each conjugate acid-base pair, identify the...Ch. 4 - Complete a net ionic equation for each...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - Prob. 4.12PCh. 4 - In acetic acid, CH3COOH, the OH hydrogen is more...Ch. 4 - Which has the larger numerical value? (a) The pKa...Ch. 4 - In each pair, select the stronger acid. (a)...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - Arrange the compounds in each set in order of...Ch. 4 - If the G for a reaction is 4.5 kcal/mol at 298 K,...Ch. 4 - Calculate the Keq for the following reactions from...Ch. 4 - Prob. 4.20PCh. 4 - Answer true or false to the following statements...Ch. 4 - In each of the following three reaction coordinate...Ch. 4 - The acid-base chemistry reaction of barium...Ch. 4 - Unless under pressure, carbonic acid (H2CO3) in...Ch. 4 - Prob. 4.25PCh. 4 - Acetic acid, CH3COOH, is a weak organic acid, pKa...Ch. 4 - Benzoic acid, C6H5COOH (pKa 4.19), is only...Ch. 4 - Prob. 4.28PCh. 4 - One way to determine the predominant species at...Ch. 4 - Will acetylene react with sodium hydride according...Ch. 4 - Prob. 4.31PCh. 4 - For each equation, label the Lewis acid and the...Ch. 4 - Complete the equation for the reaction between...Ch. 4 - Each of these reactions can be written as a Lewis...Ch. 4 - The sec-butyl cation can react as both a...Ch. 4 - Prob. 4.36APCh. 4 - Prob. 4.37APCh. 4 - Prob. 4.38APCh. 4 - Explain why the hydronium ion, H3O+, is the...Ch. 4 - What is the strongest base that can exist in...Ch. 4 - Prob. 4.42APCh. 4 - Prob. 4.43APCh. 4 - Methyl isocyanate, CH3N=C=O, is used in the...Ch. 4 - Offer an explanation for the following...Ch. 4 - Prob. 4.46APCh. 4 - Alcohols (Chapter 10) are weak organic acids, pKa...Ch. 4 - As we shall see in Chapter 19, hydrogens on a...Ch. 4 - 2,4-Pentanedione is a considerably stronger acid...Ch. 4 - Write an equation for the acid-base reaction...Ch. 4 - Prob. 4.51APCh. 4 - Prob. 4.52APCh. 4 - Prob. 4.53APCh. 4 - Following is a structural formula for imidazole, a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Answer true or false to the following statements about the mechanism of acid-base reactions. (a) The acid and base must encounter each other by a collision in order for the proton to transfer. (b) All collisions between acids and bases result in proton transfer. (c) During an acid-base reaction the lone pair on the base fills the A-H antibonding sigma orbital.arrow_forwardPredict the position of equilibrium for this acid-base reaction.arrow_forwardYou can identify relative strengths of acidity of molecules using key factors such as those in the mnemonic CARIO (Charge, Atom, Resonance, Inductivity, and Orbitals). Using this mnemonic, rank these structures in order of acidity of the indicated protons (least acidic to most acidic).arrow_forward
- Rank the Acidity of the following sets of compounds. Give an explanation for the answers.arrow_forwardExplain using examples how electronic factors (resonance and inductive effects) can be used to determine acidity and basicity of organic compounds. Use electronic and stability factors to account for the acidity of each highlighted hydrogen in the molecule below. Identify the most acidic proton. (a) HO. (b) HO. (c) H.arrow_forward1) Complete the following reactions and identify the acid, base, conjugate acid and conjugate base. CH;CH,NH2 (aq) + H2O (1) a) CH;COOH (aq) + H,O (I) b) c) Identify the conjugate base and conjugate acid of each of the following. Conjugate Base Molecule Conjugate Acid HCO3 H20 HSO4- H2PO4arrow_forward
- Would delocalization be the best explanation possible for this compound being more acidic? I was able to choose between electronegativity, inductive effect, and Resonance ( delocalization). I choose resonance because the conjugate base could share the charge on the first compound better is this the best choice. The first compound is given to be more acidicarrow_forwardWhat is the acid dissociation constant (Ka) expression for the following reaction? CH3COOH+H2O=CH3COO-+H3Oarrow_forwardWrite the expression for the acid dissociation constant (Ka), and use the value of Ka as a predictor of acid strength.arrow_forward
- Arrange the following compounds based on their increasing acidity. Write 1 for weakest acid, 2 for the next, then 3, then 4 and 5 for strongest acid,arrow_forwardConsider cyclohexane and benzene (draw the structures below). Draw the conjugate base of each molecule and then explain why cyclohexane is the weaker acid.arrow_forwardWithout reference to a pKa table, decide which compound in each pair is the stronger acid. (see attachment)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

ENVIRONMENTAL POLLUTION; Author: 7activestudio;https://www.youtube.com/watch?v=oxtMFmDTv3Q;License: Standard YouTube License, CC-BY