
Determine the point groups for
a. Ethane (staggered conformation)
b. Ethane (eclipsed conformation)
c. Chloroethane (staggered conformation)
d. 1,2-Dichloroethane (staggered anti conformation)
(a)
Interpretation:
The point group for Ethane with staggered conformation is to be determined.
Concept Introduction :
A point group defines all the operations of symmetry that could be performed on a molecule, results in an indistinguishable conformation from the original.
Answer to Problem 4.1P
The point group of ethane with staggered conformation is D3d.
Explanation of Solution
The structure of ethane in staggered conformation can be drawn as
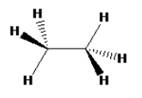
Ethane with staggered conformation has a C3 rotational axis that passes through C-C bond. There are 3 C2 rotational axes that will be perpendicular to the C3 rotational axis. It passes through the mid point of the C-C bond and doesnot have any
(b)
Interpretation: The point group for Ethane with eclipsed conformation is to be determined.
Concept Introduction:
A point group defines all the operations of symmetry that could be performed on a molecule, results in an indistinguishable conformation from the original.
Answer to Problem 4.1P
The point group of ethane with eclipsed conformation is D3h.
Explanation of Solution
The structure for ethane in eclipsed conformation is
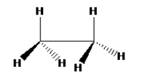
C3-rotational axis that passes through C-C bond is present in ethane in eclipsed conformation. This conformation has three C2 axes which are perpendicular to the principal C3 rotational axes and it passes through the mid-point of the C-C bond. The point group of the eclipsed ethane molecule is D3h as it has a plane of
(c)
Interpretation: The point group for chloroethane with staggered conformation is to be determined.
Concept Introduction : A point group defines all the operations of symmetry that could be performed on a molecule, results in an indistinguishable conformation from the original.
Answer to Problem 4.1P
The point group in chloroethane with staggered conformation is Cs.
Explanation of Solution
The structure of Chloroethane with staggered conformation can be drawn as
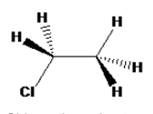
The staggered conformation of chloroethane do not have any rotational axis.
(d)
Interpretation:
The point group for 1,2-dichloroethane with staggered anti-conformation is to be determined.
Concept Introduction:
A point group defines all the operations of symmetry that could be performed on a molecule, results in an indistinguishable conformation from the original.
Answer to Problem 4.1P
The point group of 1,2-dichloroethane with staggered anti-conformation is C2h.
Explanation of Solution
The staggered anti-conformation of 1,2-dichloroethane has C2 rotational axis and it passes through the mid-point of C-C bond. This is perpendicular to the plane of Cl-C-C-Cl. The point group of the molecule is C2h as it has only one
Want to see more full solutions like this?
Chapter 4 Solutions
Inorganic Chemistry
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Chemistry: Structure and Properties
Chemistry: A Molecular Approach (4th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: The Central Science (13th Edition)
Essential Organic Chemistry (3rd Edition)
- Structural isomers can have very different point groups. Determine the point groups of 1,4cyclohexadiene and 1,3cyclohexadiene, which both have the molecular formula C6H8.arrow_forwardDetermine the point groups of the following molecules. a Hydrogen peroxide, H2O2 it is not planar. b Allene, CH2=C=CH2 c D-alanine Its absolute stereochemistry can be found in any good organic or biochemistry textbook. d L-alanine, and compare with part c above.arrow_forwardDetermine the point groups of the following molecules. a Hydrogen selenide, H2Se b Partially deuterated hydrogen sulfide, or HDS c The chair conformer of cyclohexane, C6H12 d The boat conformer of cyclohexane, C6H12arrow_forward
- Which of the following would be expected to have a dipole moment of zero on the basis of symmetry? Explain. a. SOCl2; b. SiF4; c. OF2.arrow_forward3. Assign each molecule or ion to its proper point group. a. 1,2-dibromoethane (in the gauche conformation) b. 1,2-dibromoethane (in the eclipsed conformation) C. 1-bromo-2-chloro-1,2-difluoroethane (in its most symmetric conformation) d. naphthalenearrow_forward1. Use the point group assignment flow chart to identify the point group to which each of the following species belongs. (Be sure you can actually identify these, as you'll need to be able to do so on the exam.) a. PH₂ b. PFs c. IFs d. SeF4 e. CF₂Cl₂arrow_forward
- Draw a Lewis structure and determine the point group for each of the following molecules. Indicate formal charges for ions. Indicate 3D stereochemistry by drawing solid and hashed bonds. a. XeO3 b. SOF4 c. ClOF2+ d. XeO2F2 e. TeF42-arrow_forwardWhich is most correct statement for chiral molecule? Select one: A. A molecule without plane of symmetry. B. A molecule without S - operation. O C. A molecule without carbon with four different groups. D. A molecule without any symmetry element. E. A molecule with center of inversion.arrow_forward( find point group of each molecule with detail explanationarrow_forward
- The above three Ferrocenes are...............: of the same point group diastereomers of different chemical formulas conformations 20 b Fearrow_forward3. Assign point group to each of the following molecules: (a) NF3, (b) H2O2, (c) SF3C1arrow_forwardDetermine the point group of the molecule shown. Be sure I CI OF to rotate the molecule so that you can see all of it. Your answer should have the correct formatting for point groups (e.g., D4h). F point group: CI F F F. F.arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

