
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.17C, Problem 2.26P
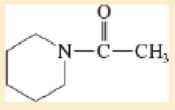
Draw a Lewis structure and classify each of the following compounds.
- a. CH3CH2CONHCH3
- b. (CH3CH2)2NH
- c. (CH3)2CHCOOCH3
- d. CH3CHCHCOCl
- e. (CH3CH2)2O
- f. CH3CH2CH2CN
- g. (CH3)3CCH2CH2COOH
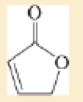
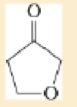
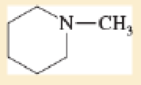
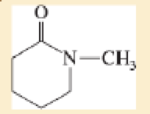
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a Lewis structure for each ion.
a. CH3O−
b. HC2−
c. (CH3NH3)+
d. (CH3NH)−
3. Draw the best complete Lewis structure for each of the following
molecules or ions. Make sure to include all lone pairs and formal
charges when necessary
a. CO₂H2
b. CH3O-
c. CNH6+
A. CHF
i. Best Lewis Structure
B. HNO (H is connected to one of the O's)
i. Best Lewis Structure
ii. Electron geometry on the C atom
ii. Electron geometry on the N atom
iii. Approximate bond angles about the C atom
iii. Approximate bond angles around the N atom
v. Draw the shape with in and out wedges (as necessary) and
dipole arrows around the C atom.
v. Draw the shape with in and out wedges (as necessary) and
dipole arrows around the N atom.
vi. Is the molecule polar or nonpolar?
vi. Is the molecule polar or nonpolar?
Chapter 2 Solutions
Organic Chemistry (9th Edition)
Ch. 2.1A - Prob. 2.1PCh. 2.1B - The NF bond is more polar than the NH bond: but...Ch. 2.1B - For each of the following compounds 1. Draw the...Ch. 2.1B - Two isomers of 1,2-dichloroethene are known One...Ch. 2.2C - Prob. 2.5PCh. 2.2C - Prob. 2.6PCh. 2.3 - Prob. 2.7PCh. 2.4 - Calculate the pH of the following solutions a....Ch. 2.6A - Ammonia appears in Table 2-2 as both an acid and a...Ch. 2.7 - Write equations for the following acid-base...
Ch. 2.7 - Ethanol, methylamine. and acetic acid are all...Ch. 2.8 - Prob. 2.12PCh. 2.10 - Write equations for the following acid-base...Ch. 2.10 - Rank the following acids in decreasing order of...Ch. 2.11 - Prob. 2.15PCh. 2.11 - Prob. 2.16PCh. 2.11 - Consider each pair of bases and explain which one...Ch. 2.12 - Which is a stronger base ethoxide ion or acetate...Ch. 2.12 - Prob. 2.19PCh. 2.12 - Prob. 2.20PCh. 2.12 - Prob. 2.21PCh. 2.12 - Choose the more basic member of each pair of...Ch. 2.14 - Prob. 2.23PCh. 2.15D - Classify the following hydrocarbons and draw a...Ch. 2.16D - Prob. 2.25PCh. 2.17C - Draw a Lewis structure and classify each of the...Ch. 2.17C - Circle the functional groups in the following...Ch. 2 - The CN triple bond in acetonitrile has a dipole...Ch. 2 - Prob. 2.29SPCh. 2 - Sulfur dioxide has a dipole moment of 1.60 D....Ch. 2 - Which of the following pure compounds can form...Ch. 2 - Predict which member of each pair is more soluble...Ch. 2 - Prob. 2.33SPCh. 2 - Prob. 2.34SPCh. 2 - Predict which compound in each pair has the higher...Ch. 2 - All of the following compounds can react as acids...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - The Ka of phenylacetic acid is 5 2 105, and the...Ch. 2 - The following compound can become protonated on...Ch. 2 - The following compounds are listed in increasing...Ch. 2 - Prob. 2.42SPCh. 2 - Prob. 2.43SPCh. 2 - Compare the relative acidity of 1-molar aqueous...Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - The following compounds can all react as bases. a....Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - Prob. 2.48SPCh. 2 - Methyllithium (CH3Li) is often used as a base in...Ch. 2 - Label the reactants in these acid-base reactions...Ch. 2 - In each reaction, label the reactants as Lewis...Ch. 2 - Prob. 2.52SPCh. 2 - Each of these compounds can react as a nucleophile...Ch. 2 - Prob. 2.54SPCh. 2 - Give a definition and an example for each class of...Ch. 2 - Circle the functional groups in the following...Ch. 2 - Prob. 2.57SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the Lewis structure for each organic compound from its condensed structural formula. a. CH4 b. CH,NH2 с. НСНО d. CH,CH2OH е. НСООНarrow_forwardDraw a valid Lewis structure for each species.a. CH3CH3b. CH5Nc. CH3Clarrow_forwardDraw a valid Lewis structure for each compound. a. Ng b. (CH,OH)* (CH;CH2) d. HNNH е. НаBN C.arrow_forward
- Draw the Lewis structure for each organic compound from its condensed structural formula.a. C3H8 b. CH3OCH3 c. CH3COCH3d. CH3COOH e. CH3CHOarrow_forwarda. Draw two Lewis structures for C2H6O. b. Draw three Lewis structures for C3H8O.arrow_forwardH H- Н Н H:0: НН Н Н H 4-5 H H/ Н нн Н 0: д H Н H H Н d. b. EHO HO(HO HO HO HO HO H H Т нн Н н н нн H H H О н н H H H Hн Н O H H. 'Н Which of the following structures represents the correct Lewis structure for the molecule below? a.arrow_forward
- 1. What is the relationship between the following compounds? H :0: H H:0: H H-C=C-C-C-C-H H-C-C-C=Ċ-H H HH H. H-C-H B. constitutional isomers C. the same structure A. isotopes D. composed of different elements E. no relationship 2. What is the correct Lewis structure for HN3, including the formal charges? CIHarrow_forward2. What type of chemical bonding is present in the following compound? CH3CH2CH2CH2Br O a. A O b. B O C. C O d. D A) Polar covalent B) Hydrogen C) lonic D) polar covalent and non-polar covalentarrow_forward1. NH2Cl a. In NH2Cl there is a total of ____________ valence electrons. b. The NH2Cl model consists of how many lone pairs of electrons c. How many bonds are in the NH2Cl molecule? d . What is the shape the atoms make (excluding lone pairs) in NH2Cl?arrow_forward
- 1. Draw the most appropriate Lewis structure(s) for CCI4. What is the electron domain geometry an- the molecular geometry? Does the molecule have a dipole?arrow_forward1. Convert the condensed formulas to Lewis structures (show all atoms, bonds, lone pairs) a. CH3(CH₂)4OH b. CH3(CH₂)4CH(CH3)2 C. (CH3)2CHO(CH2)4CH3 d. (CH3)2CHCHOarrow_forwardWhat is the lewis structure for the following 1. CH3C(CH3)2CH3 2 CH3CH2CH2CH2CH2CH2CH3 3. CH3CH2CH2CH2CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY