Q: A sticky substance that upon dissolving in water, is found to be a nonelectrolyte, has a concentrati...
A: Molarity=Number of moles of solute/volume of soln in Lmolality=no. of moles of solute/mass of solven...
Q: If energy is required to form monatomic ions from metals and nonmetals, why do ionic compounds exist...
A: Ionic compounds are exist as crystal lattice and as not simple ion pairs.
Q: A 0.16-g sample of a purified protein is dissolved in water to give 2.0 mL of solution. The osmotic ...
A: Osmotic pressure for dilute solution, π = CST π = nVST where, π = Osmotic pressure C = concentrati...
Q: (a) Determine the oxidation number of lead in each of the following oxides: PbO, PbO2, Pb2O3, Pb3O4....
A: Oxidation number is defined as the number that is assigned to an atom when it loses or gains electro...
Q: How many kilojoules are released when 8.2 g of water condenses at 100°C and cools to 15 degrees Cels...
A: Given , 8.2 g of water condenses at 100°C and cools to 15 degrees Celsius m= 8.2 g Ti= 100°C Tf=15°C...
Q: The balanced equation for the decomposition of water is shown below. 2 H2O → 2 H2 + O2 If 0.72 g of ...
A: SO ACCORDING TO THIS BALANCED EQUATION 36 GRAMS OF WATER WILL DECOMPOSE INTO 4 GRAMS OF HYDROGEN GAS...
Q: Which of the following represents the correct balanced chemical equation for the burning of coal whi...
A: When coal is burned, it reacts with the oxygen in the air. This chemical reaction converts the store...
Q: how many moles are in 46.5 g of pure aluminum and how many atoms are in 46.5 g of pure aluminum?
A: 1. Mole in terms of mass: 1 mole = atomic mass of an atom. No. of moles (n) of an atom can be calcu...
Q: 204 + Dye → 2 CO2 + Excited Dye wo glow sticks are placed in water baths, one in hot water and the o...
A: Collision theory is used to predict the rates of chemical reaction particularly for gases. It is bas...
Q: Please answer everything pooooo this can really help me :(
A: "Since you've asked multiple questions, we'll solve the first three parts for you and for the rest t...
Q: Ammonia gas can be prepared by the following reaction: CaO(s) +2 NH, Cl(s) → 2 NH3 (g) +H20(g) + CaC...
A:
Q: What is the line notation for each compound? ethanol methanol acetone pentane 1-propanol 1-butanol
A: Line notations represent structures as a linear string of characters. The Simplified Molecular-Input...
Q: Iron(III) oxide and hydrogen react to form iron and water, like this: Fe,O3(s)+3 H,(g)–2Fe(s)+3H,O(g...
A: A numerical problem based on equilibrium concept, which is to be accomplished.
Q: A patient needs 1.5 g of a solid must be given to the patient pe O 60 tablets
A: The dose of a drug: it is the quantitative amount taken by patient for the intended medicinal effect...
Q: Calculate the freezing point depression of naphthalene/biphenyl Find the molality of Naphthalene/...
A:
Q: 1. State the functional groups present. What suffix will be used in the IUPAC name for this compounc...
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any ...
Q: Write the chemical composition of the compound formed when ferric chloride is added containing both ...
A: The chemical formula of Ferric chloride is FeCl3 . Ferric chloride contains halogen i.e., chloride i...
Q: 8. Predict the major product for each reaction of triglycerides. These are not new reactions! You le...
A: Hydrogenation is the process of adding hydrogen atoms to unsaturated fats until it becomes saturated...
Q: An automobile engine provides 576 Joules of work to push the pistons and generates 2340 Joules of he...
A:
Q: To improve conductivity in the electroplating of automo-bile bumpers, a thin coating of copper separ...
A: a) The mass of copper has to be determined, The half-reaction can be written as, Cu2+(aq)+2e-→Cu(s) ...
Q: What is the number of formula units in a 9.72 mol sample of NH4NO3?
A: 1 mole of the sample contains 6.02 x 10^23 formula units
Q: I need help on 3 f-h
A:
Q: A solution contains 3.90 g of a nonvolatile pure hydrocarbon in 95 g acetone. The boiling points of ...
A:
Q: Question attached
A: Retrosynthesis: The retrosynthesis is a backwards synthesis of target molecule from simpler precurso...
Q: N2 (g) + O2 (g) =2 NO (g) Describe a step-by-step method to calculate the equilibrium concentration ...
A:
Q: Chemistry Question
A: In water, the sodium carbonate dissociates into two distinct stages. ...
Q: The following reactants will undergo a Robinson annulation reaction in the presence of base (KOH). D...
A:
Q: QUESTION 7 The following reaction is exothermic: CF2Cl2() - CF2Cg) Cg) Select the energy diagram tha...
A: The heat is released during the exothermic process, therefore the energy of the reactants in this pr...
Q: Which resonance form contributes more to the resonance hybrid? Explain briefly. N-H H- CH3 H-ö CH3 W...
A: Since you have posted a question with multiple subparts, we will solve the first three subparts for ...
Q: Where are the nodes in the wavefunction for a particle confined to a box with 0 < x < a and n=...
A: Wave function for a particle in a box with 0 < x < a and n=3 No of nodes = to be determined. ...
Q: Calculate [OH−] for the following solution; [H3O+] = 8.87×10−9 M Is the solution basic at 25°C?
A: pH is negative logarithm of hydronium ion. Given, [H3O+] = 8.87×10−9 M
Q: I need full accurate and detailed answers.
A: Arrhenius equation is an expression that relates to the activation energy, temperature, and the rate...
Q: Write the steps of ‘Modern Adsorption Theory of Heterogenous Catalysis.’
A: The Modern Adsorption Theory can be well explained the overall basic steps involved in the heterogen...
Q: (S)-Adenosylmethionine (SAM) is a cosubstrate that is involved in biological methyl group transfers....
A: (a) The appropriate curved arrows for the given reaction are shown below:
Q: D) Explain your answer. This is the most important part. Please type on an attached page. A complete...
A: Boiling point direct proportional to intermolecular force.
Q: I need help with answering the second part of this problem.
A: Conformational isomers are those isomers which are formed by the free rotation around Carbon Carbon ...
Q: If the percent yield for the following reaction is 65.0%, how many grams of KClO3 are needed to prod...
A: The question is based on the concept of reaction Stoichiometry. we have been given a reaction and pe...
Q: What volume of ethylene glycol (C2H6O2), a nonelectolyte, must be added to 14.0 L water to produce a...
A: ∆Tf = Kf x Molality Molality = Number of moles of solute/Mass of solvent in Kg
Q: QUESTION 10 Measurements taken during the following reaction showed a concentration of carbon m...
A:
Q: Carbon-hydrogen bonds exhibit a range of different chemical reactivity that depends on molecular str...
A: The carbon atoms of organic compounds are classified by the number of other carbons directly bonded ...
Q: Sodium and chlorine react to form sodium chloride:2 Na(s) + Cl2( g)¡2 NaCl(s)What is the theoretical...
A: The limiting reagent in a chemical reaction is a reactant that is totally consumed when the chemical...
Q: You start the following reaction with 0.411 M N2 and 0.398 M H2. At equilibrium, you determine the c...
A:
Q: Write the structural formula and name of the organic product for the reaction between an alkyne and ...
A:
Q: Question attached
A: Given: The mass of glucose is 80 g. The mass of the solvent is 2Kg. We know that the molecular weigh...
Q: Complete and balance the following nuclear equations by supplying the missing particle: 1 1H + 11 5B...
A: The total mass number and proton: H11+B511,mass number=11+1=12The number of proton=5+1=6
Q: A sample of 3.929 g of polystyrene was dissolved in toluene and diluted to a total volume of 100.8 m...
A:
Q: How many peaks are present in the NMR signal of attached labeled proton?
A: Number of peaks correspond to the number of different type of protons present in the molecule.
Q: The protein ribonuclease A in its native, or most stable, form is folded into a compact globular sha...
A:
Q: A sheet of gold weighing 10.0 g and at a temperature of 18.0°C is placed flat on a sheet of iron wei...
A:
Q: Q13.58) How much dry solute would you take to prepare each of the following solutions from the dry s...
A: there are many concentration measurement terms such as molarity, molality and normality etc.
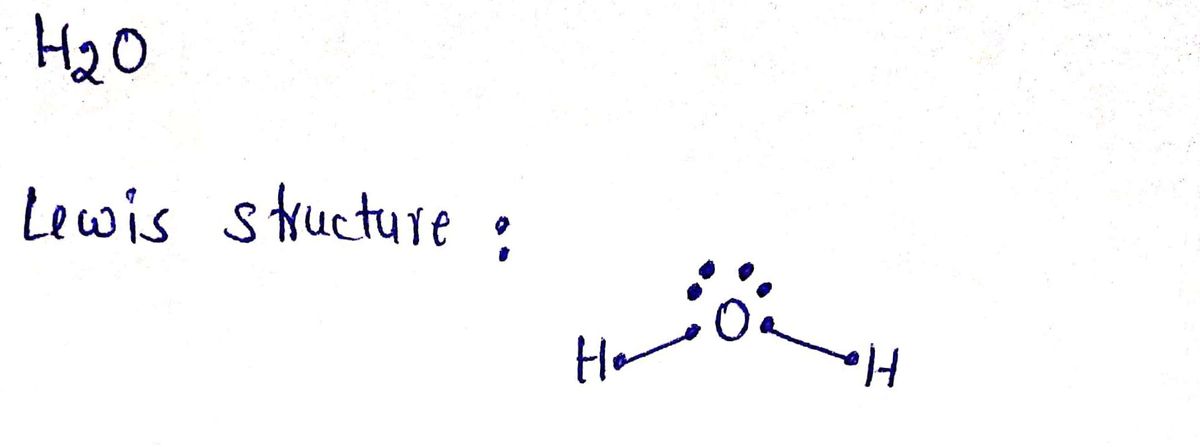
- Draw the lewis structures for H₂O, NH₃, CH₄
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Help me write the Lewis symbol for (Rn) please10.) The structural formula of a certain aldehyde (related to formaldehyde) is H3C-CH2-CHO. Draw a Lewis structure for this aldehyde and determine the number of bonds present. Note that a single or a double or a triple bond counts as one bond. Write the number, not the word.Write the Lewis symbols that represent the ions in ionic compound. RbF



