Carbon monoxide [CO] exhibits an IR absorption at 2143cm-1, acetone [CH3C(O)CH3],exhibits an IR absorption for the CO vibration at 1715 cm-1 and ethanol [CH3CH2OH] exhibits an IR absorption for CO at ~1150 cm-1. Draw Lewis structures of these three molecules and use your structures to explain the observed differences in the CO IR absorption peak frequency (nCO).
Carbon monoxide [CO] exhibits an IR absorption at 2143cm-1, acetone [CH3C(O)CH3],exhibits an IR absorption for the CO vibration at 1715 cm-1 and ethanol [CH3CH2OH] exhibits an IR absorption for CO at ~1150 cm-1. Draw Lewis structures of these three molecules and use your structures to explain the observed differences in the CO IR absorption peak frequency (nCO).
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter20: Dienes, Conjugated Systems, And Pericyclic Reactions
Section20.3: Uv-visible Spectroscopy
Problem 20.5P
Related questions
Question
Carbon monoxide [CO] exhibits an IR absorption at 2143cm-1, acetone [CH3C(O)CH3],exhibits an IR absorption for the CO vibration at 1715 cm-1 and ethanol [CH3CH2OH] exhibits an IR absorption for CO at ~1150 cm-1. Draw Lewis structures of these three molecules and use your structures to explain the observed differences in the CO IR absorption peak frequency (nCO).
Expert Solution
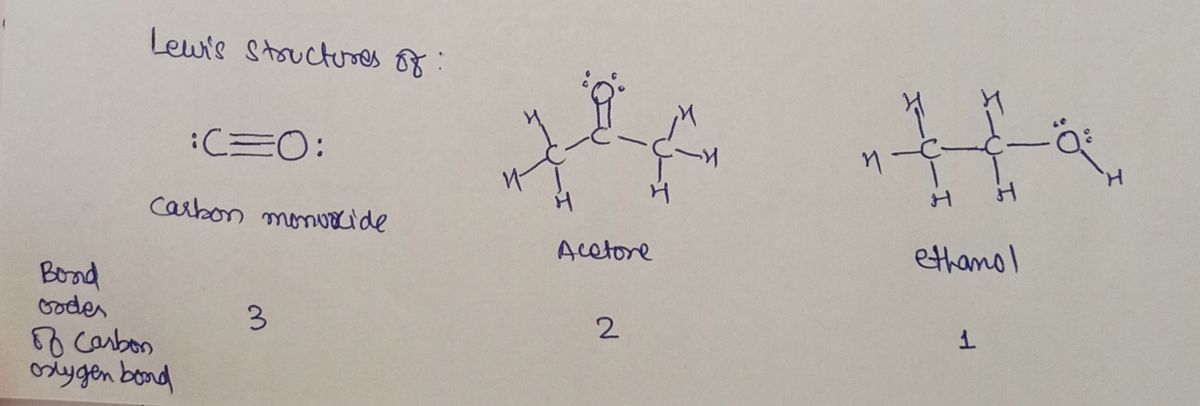
Lewis structures:
Lewis structure of the compounds with Carbon and oxygen bond, bond order is shown below:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning